NSRC Events
NSRC Workshops
The NSRCs hold joint workshops to share research and user projects that are ongoing at the five centers. These exchanges of information have provided the staff at the NSRCs with the opportunity to learn about topics/thrusts in nanoscience at the other nanocenters and to develop an understanding of the different areas of expertise among the staff members. They have also facilitated discussions towards possible future areas of collaboration between the centers and provided basic information so that potential NSRC users can be directed toward the optimal center and staff to meet their research needs.
CFN - 84th Physical Electronics Conference
Upton, NY 11-Aug-2025 – 14-Aug-2025

Annual User Meeting Dates
CFN - NSLS-II & CFN Users' Meeting
Upton, NY 28-Apr-2025 – 30-Apr-2025
CINT - 2025 CINT User Meeting
Santa Fe, NM 15-Sep-2025 – 16-Sep-2025
CNMS - 2014 User Meeting (Registration deadline: September 3, 2014)
Oak Ridge, TN 15-Sep-2014 – 19-Sep-2014
CNMS - 2022 CNMS User Meeting
(Virtual) Oak Ridge, TN 8-Aug-2022 – 12-Aug-2022
CNM - 2025 APS/CNM Users Meeting
5-May-2025 – 9-May-2025
TMF - 2014 User Meeting
Berkeley, CA 25-Aug-2014 – 26-Aug-2014

News and Highlights
Current Highlights
Computational Design of a New MOF for N2/CH4 Separations
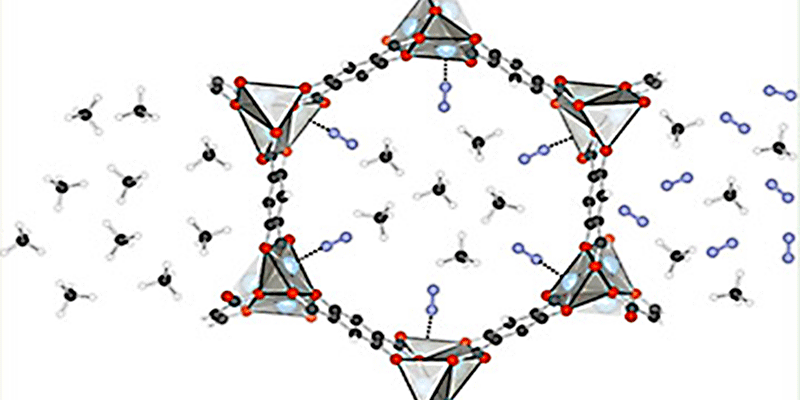
A collaborative team of Molecular Foundry Users and staff used computation to design and predict a new metal–organic framework (MOF) able to separate dinitrogen from methane and other methane-rich gases.
2D MoS2 Templates Unlock High-Performance Perovskite Solar Cells
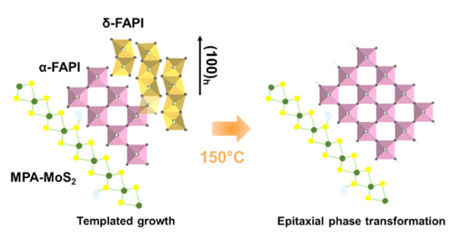
Scientific Achievement
Using functionalized 2D molybdenum disulfide (MoS2) nanosheets, researchers from Stony Brook University (SBU), The City University of New York (CUNY), and Center for Functional Nanomaterials (CFN) templated growth of the photoactive perovskite phase, boosting both solar cell efficiency and stability.
Significance and Impact
The work reveals a new mechanism by which 2D additives control crystallization and boost hybrid perovskite performance for solar energy conversion and optoelectronics applications.
Advanced Electron Microscopy Reveals Processing Impacts on Quantum Circuits
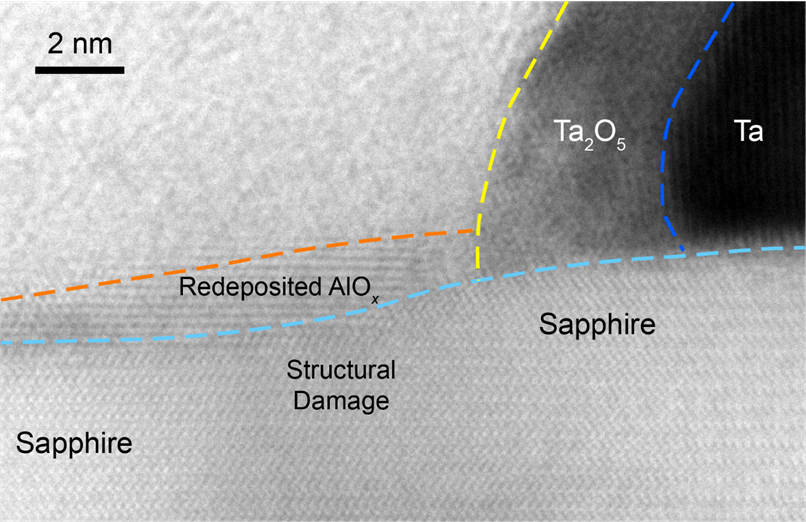
Scientific Achievement
Researchers from the Co-design Center for Quantum Advantage (C2QA) investigated how wet and dry etching processes affect the morphology, chemistry, and strain in high-Q tantalum resonators, currently the most promising materials for high-performance superconducting qubits.
Significance and Impact
Processing-induced effects, such as lattice deformations and substrate damage, can influence the performance of superconducting quantum devices. Understanding these effects and their impact helps optimize device fabrication.
Atomically Thin Mirrors: A New Frontier in Light Confinement
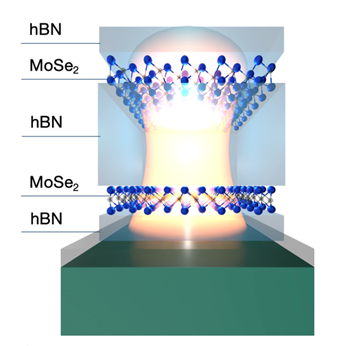
Scientific Achievement
Scientists from the University of Maryland, working with Center for Functional Nanomaterials staffs, developed a subwavelength 2D nanocavity using atomically thin transition metal dichalcogenide (TMD) mirrors with unique excitonic properties, enabling confined and tunable optical modes.
Significance and Impact
This work establishes a new platform for nanoscale light manipulation using atomically thin materials, with potential use in quantum information processing, optical computing, and quantum photonics.
Archived News
Nanometer-scale mapping of irreversible electrochemical nucleation processes on solid Li-ion electrolytes.

Nanoparticles of lithium metal formed on the surface of a solid state lithium ion electrolyte by an atomic force microscope. The particle size and height can be controlled by using carefully chosen voltage amplitude and sweep rates. The particles can be as small as 50 nanometers in diameter and a few nanometers high, and can potentially be used in lithium nanobatteries. A. Kumar, T.M. Arruda, A. Tselev, I.N. Ivanov, J.S. Lawton, T.A. Zawodzinski, O. Butyaev, X. Zayats, S. Jesse, and S.V. Kalinin, “Nanometer-scale mapping of irreversible electrochemical nucleation processes on solid Li-ion electrolytes.” Scientific Reports 3, 1621 (2013)
Platinum-Lead Core/Shell Nanoplates are High-Performance, Highly-Stable Fuel Cell Catalysts
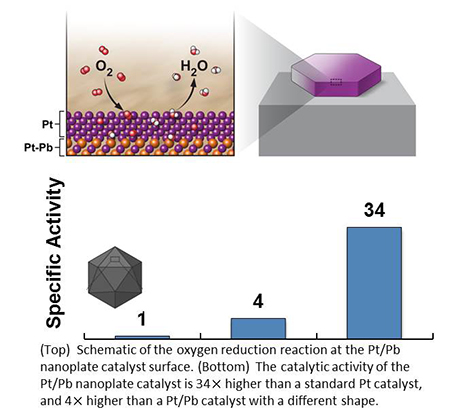
A team of scientists from CFN, Peking University, and Soochow University designed and characterized a new fuel cell catalyst — a platinum-lead core/shell structure, shaped as a nanoplate. The catalyst shape and chemical composition dramatically enhances the oxygen evolution reaction — important for fuel cell performance — while providing stability during operation.
Screening for Disease or Toxins in a Drop of Blood


Foundry industry users developed a multinozzle emitter array (MEA), a silicon chip that can dramatically shorten the time it takes to identify proteins, peptides, and other molecular components within small volumes of biological samples. This patented technology is now being commercialized by Newomics Inc., to further develop the product and build a platform for personalized health care. Some of the early work on multinozzle emitters was done at the Molecular Foundry.
Newomics’ product, which is based on the core technology developed at Berkeley Lab, is designed to work with mass spectrometers, a machine commonly used by research scientists, the pharmaceutical industry, and increasingly in clinical labs, to measure the structure and concentration of molecules. Once molecular parts are isolated, scientists can begin to understand how they work together as a system, a field known as systems biology, which holds great promise for better medicines and diagnostics as well as a host of other applications.
The dominant method for analyzing biological molecules such as peptides and proteins in a complex mixture is electrospray ionization mass spectrometry (ESI-MS), a technique in which molecular samples are delivered to the machine as an ionized mist, propelled by an electric current. But there is a bottleneck at the front end of ESI-MS, making it slow and expensive. Each sample has to be loaded, lined up, and sprayed one at a time.
Instead of a single capillary, Newomics’ M3 emitter has eight or more nozzles working together to split a single large flow into smaller flows. For the MEA, up to 96 M3 emitters are packaged on a single chip. The development of these technologies involved a blend of microfluidic, microelectronic, and electrochemical innovations.
By clearing up the bottleneck and increasing throughput, Newomics’ emitter could dramatically reduce the cost of testing each sample. And by improving the sensitivity, it will also be possible to detect very low concentrations of molecules. For example, they showed they could analyze many different modified forms of proteins such as glycated albumin and apolipoproteins, in addition to the conventional glucose and HbA1c in diabetes monitoring, using a single drop of blood. Such tests have the potential to enable better long-term monitoring and disease management of diabetes.
Data Science Heals Imperfect X-ray Scattering Datasets
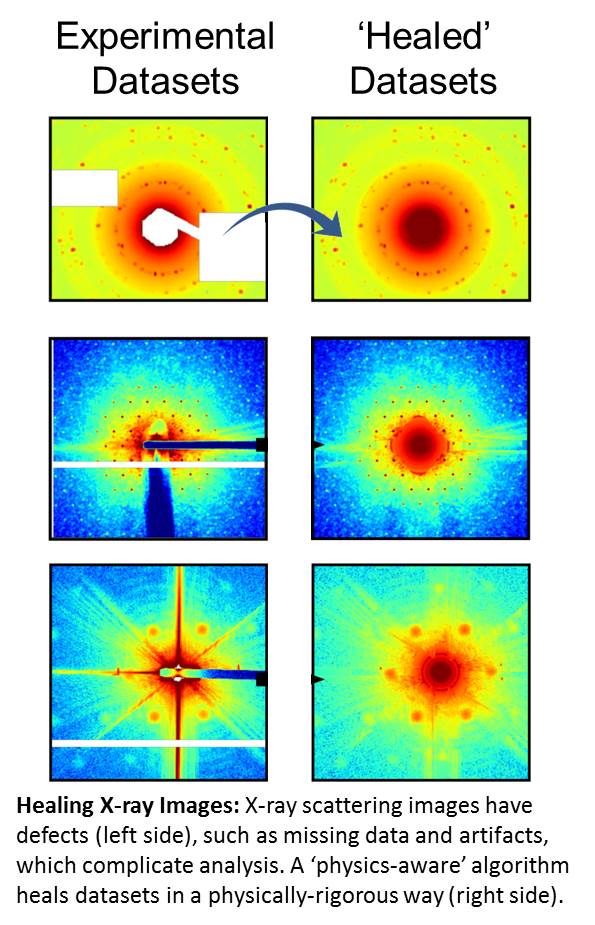
The team of scientists developed and implemented a ‘physics-aware’ algorithm to correct for missing information in experimental X-ray scattering datasets. Because the algorithm relies on well-understood physics of X-ray scattering, the ‘healing’ operation provides robust and physically-rigorous results and outperforms all other conventional image interpolation methods.
Experimental X-ray scattering images always contain missing data and artifacts, which complicate further analysis, especially rapid, automated analysis. This healing operation is an essential pre-processing step for machine-learning interpretation of scientific data.
X-ray scattering is a powerful way to measure the structure of materials at the molecular- and nano-scale. Scattering images contain features, such as peaks and rings, which encode structural information. As with most scientific data, collected X-ray scattering images are inevitably ‘incomplete,’ with missing data being due to limits of the measurement, or experimental considerations. These missing data render automated data analysis of the datasets much more difficult. In this work, the team developed an image healing algorithm designed for X-ray scattering/diffraction datasets. Because the algorithm is ‘physics-aware’ (incorporating known properties of an X-ray scattering measurement), it outperforms all other image healing methods when applied to X-ray scattering data. The healed images can then be easily fed into existing data analysis pipelines. Importantly, the image healing is also a crucial pre-processing step for input to machine-learning methods — which would otherwise tend to focus on the high-intensity — but ultimately irrelevant — image defects.
New Catalyst Gives Artificial Photosynthesis a Big Boost


Researchers have created a new catalyst that brings them one step closer to artificial photosynthesis — a system that would use renewable energy to convert carbon dioxide (CO2) into stored chemical energy.
As in plants, their system consists of two linked chemical reactions: one that splits water (H2O) into protons and oxygen gas, and another that converts CO2 into carbon monoxide (CO). The CO can then be converted into hydrocarbon fuels through an established industrial process. The system would allow both the capture of carbon emissions and the storage of energy from solar or wind power.
Foundry scientists Yufeng Liang and David Prendergast performed theoretical modeling work used to interpret X-ray spectroscopy measurements made in the study, published Nov. 20 in Nature Chemistry. This work was done in support of a project originally proposed by a team from the University of Toronto.
Last year, the team developed catalysts for such reactions. But while one of their catalysts worked under neutral conditions, the other required high pH levels in order to be most active. That meant that when the two were combined, the overall process was not as efficient as it could be: energy was lost when moving charged particles between the two parts of the system.
The team has now overcome this problem by developing a new catalyst for the first reaction – the one that splits water into protons and oxygen gas. Unlike the previous catalyst, this one works at neutral pH, and under those conditions it performs better than any other catalyst previously reported.
The new catalyst is made of nickel, iron, cobalt and phosphorus, all elements that are low-cost and pose few safety hazards. It can be synthesized at room temperature using relatively inexpensive equipment, and the team showed that it remained stable as long as they tested it, a total of 100 hours.
The team employed X-ray experiments at the Canadian Light Source and Berkeley Lab’s Advanced Light Source (ALS) to reveal the working principle behind this new catalyst, mainly focusing on the nickel chemistry during the reaction itself. The Theory Facility at Berkeley Lab’s Molecular Foundry specializes in the interpretation of such X-ray results, connecting chemical intuition to the atomic and electronic structure models of working materials.
Sorting molecules with self-assembled nanoscale membranes
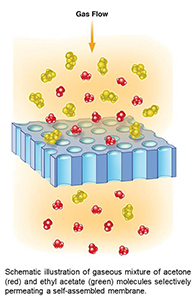
What is the scientific achievement?
We have fabricated highly-porous, highly-uniform silicon nitride membranes by replicating features from self-assembled block copolymer films. With porosities over 30% and thickness <100 nm, the membranes are designed for high throughput. Pore sizes are controllably tuned to molecular scales, for selective gas permeation. Capillary condensation within nanoscale pores enhances selectivity beyond that expected from molecule size differences.
Why does this achievement matter?
Membranes underlie integral separation processes in energy production, water purification, medicine, environmental cleanup, and chemical processing. These highly-uniform, highly-porous inorganic membranes may provide durability for high temperature operation in extreme environments.
Getting an Up-Close, 3D View of Optical Nanomaterials

What is the scientific achievement?
CFN users from Rutgers University worked with CFN staff to perform high-resolution, 3D imaging of metallic nanostructures by scanning transmission electron microscopy (STEM). The measured 3D structure of these ‘nanostars’ was used as input for finite element simulations of the material physical and optical properties, in remarkable agreement with experimental measurements.
Why does this achievement matter?
Nanomaterials can have enhanced optical properties stemming from plasmonic effects — giving them promise for advanced sensors and diagnostic applications. This study represents the first time that information from STEM tomography has been used to predict nanomaterial physical and optical properties.
Deactivation Mechanism in NOx Reduction Zeolite Catalysts Revealed by Atom Probe Tomography

Atom-level deactivation processes in industrial zeolite catalysts are revealed in atom probe tomography (APT), which yields the first direct observations of chemical distributions. J. E. Schmidt, R. Oord, W. Guo, J. D. Poplawsky, B. M. Weckhuysen.
Nature Communications 8, 1666 (2017). DOI: 10.1038/s41467-017-01765-0
Imaging Flexible DNA 'Building Blocks' in 3-D


Over the past decade, researchers have been working to create nanoscale materials and devices using DNA as construction materials through a process called “DNA origami.”
Now, for the first time, a team of researchers from Lawrence Berkeley National Laboratory (Berkeley Lab) and Ohio State University have generated 3-D images from 129 individual molecules of flexible DNA origami particles. Their work provides the first experimental verification of the theoretical model of DNA origami.
The research team focused on DNA structures modeled after a basic mechanism called a “Bennett linkage,” which is a 3-D structure consisting of a chain of four rods connected by hinges. This creates a skewed quadrilateral shape in which the hinges are not parallel or in-line. Using Bennett linkages as building blocks, it’s possible to create expandable, useful structures, like supports for tents that can be rapidly assembled.
The researchers relied on a technique developed at the Molecular Foundry to image the individual molecules that make up these structures. The method, called individual-particle electron tomography (IPET), takes pictures of a target molecule from multiple viewing angles, and then combines these pictures to create one 3-D, whole-molecule rendering, similar to how a medical computed tomography (CT) scan works.
Researchers captured 129 3-D images, with a resolution of 6 to 14 nanometers, that enabled them to tease out information about the dynamics and flexibility of DNA origami structures.
Self-assembly of Metal Organic Frameworks (MOFs) and Nanocrystals into Open Network 2D Structures

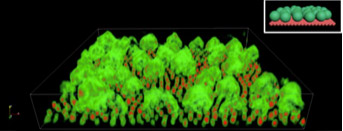
An international team of Foundry staff and users have created a method to direct the self-assembly of MOFs and nanocrystals into new types of 2D structures. This is the first time that researchers have been able to guide the self-assembly of organized 2D structures using MOFs and nanocrystals. This development will enable the design of new functional materials for catalysts, energy storage, and more.
Super-stable Platinum-alloy/Graphene-tube Catalysts
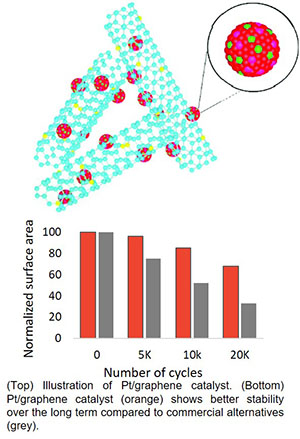
What is the scientific achievement?
A team of scientists from the CFN and the University of Buffalo developed and characterized a new Pt-alloy catalyst to promote the oxygen reduction reaction in fuel cells. This catalyst is formed by annealing Pt nanoparticles deposited onto graphene tubes co-doped with nitrogen, nickel, and cobalt. After annealing, the resulting Pt-alloy catalyst and N-doped graphene support enhanced catalytic activity and stability.
Why does this achievement matter?
Pt catalysts on carbon supports are used to promote the oxygen reduction reaction in commercial fuel cells, but these catalysts suffer from poor long-term stability. The Pt-alloy catalysts synthesized in this work have excellent activity and improved stability, and represent a new design strategy exploiting a unique hybrid configuration.
New Polymer Model Helps Explain the Origins of Life
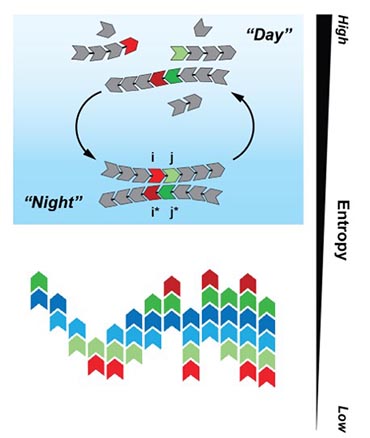
Scientific Achievement
CFN staff in collaboration with the University of Illinois at Urbana-Champaign developed a polymerization model that evolves from a "soup" of different monomers into a complex system made from a limited number of polymer segments, which reduces the entropy in the system. Numerical results, supported by mathematical analysis, confirm the survival and extinction process that resembles natural selection, and drives the dramatic decrease of informational entropy.
Significance and Impact
The emergence of life from non-living matter is one of the greatest mysteries of fundamental science. The model could also be applied to make biopolymers and nanostructures.
What is the scientific achievement?
CFN staff in collaboration with the University of Illinois at Urbana-Champaign developed a polymerization model that evolves from a "soup" of different monomers into a complex system made from a limited number of polymer segments, which reduces the entropy in the system. Numerical results, supported by mathematical analysis, confirm the survival and extinction process that resembles natural selection, and drives the dramatic decrease of informational entropy.
Why does this achievement matter?
The emergence of life from non-living matter is one of the greatest mysteries of fundamental science. The model could also be applied to make biopolymers and nanostructures.
Smaller Nanoparticles are Better Battery Materials

What is the scientific achievement?
A team of scientists from the Center for Mesoscale Transport Properties Energy Frontier Research Center working with CFN staff to study in real time how the size of zinc ferrite (ZnFe2O) nanoparticles determines their performance during battery operation. The team found that the improved performance observed when using smaller nanoparticles (6-9 nm) is because lithium is taken up by forming a solid-solution, a more efficient reaction pathway.
Why does this achievement matter?
Understanding lithiation in materials as a function of particle size is helpful in designing batteries with improved performance and longevity. In-situ, dry cell transmission electron microscopy allows direct observation of structural changes in real time and with high spatial resolution.
Small Polymers Can Have a Big Impact on Self-Assembly

What is the scientific achievement?
CFN staff members found that increasing the film thickness of cylinder-forming block copolymers frustrates their self-assembly and results in highly defective nanoscale patterns. However, blending small homopolymers into the films alleviates the frustration, especially during early stages of pattern formation. In-situ grazing-incidence X-ray scattering and ex-situ electron microscopy reveal the ways that these homopolymers help produce more uniform and well-oriented patterns.
Why does this achievement matter?
Improving the quality of nanopatterns produced via self-assembly in thicker polymer films creates new opportunities to engineer functional, large-area surface nanotextures and nanoporous membranes.
Engineered Nanowrappers Carry and Release Cargo
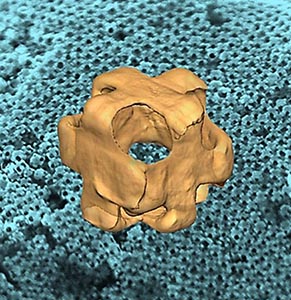
What is the scientific achievement?
CFN Staff have discovered a new method to synthesize hollow metallic nanostructures with surface openings, which can carry and deliver cargos of guest nanoobjects. These nanowrappers have unique optical signatures originating from plasmonic effects and their complex nanoarchitecture. Advanced electron tomography provides 3D images at different stages of synthesis, which tracks their transition from Ag nanocubes with sharp corners to Au-Ag alloy nanowrappers with large cubic pores at all corners.
Why does this achievement matter?
This research is a promising new strategy for synthesis of porous, 3D nanoarchitectures. Nanowrappers have biomedical potential as photothermal therapeutics, vehicles for photoinduced drug delivery, or agents for improved imaging contrast.
Data Analytics Uncovers the Structure of Ultrathin Catalyst Coatings

What is the scientific achievement?
CFN scientists and BNL collaborators from NSLS-II and the Computational Science Initiative deduced the structure of ultrathin titania (TiO2) coatings on ZnO nanowire photocatalysts by employing new data analytic approaches to X-ray absorption near edge structure (XANES) measurements. The ultrathin, amorphous TiO2 promotes efficient charge transfer during photocatalytic water splitting while also protecting the catalyst against photocorrosion.
Why does this achievement matter?
This research implements a unique, data-driven approach to deciphering the structure of highly amorphous materials at the smallest dimensions from X-ray spectra, and lays the groundwork for understanding chemical and electronic properties.
Observing the Obstacles Underlying Battery Capacity Fade
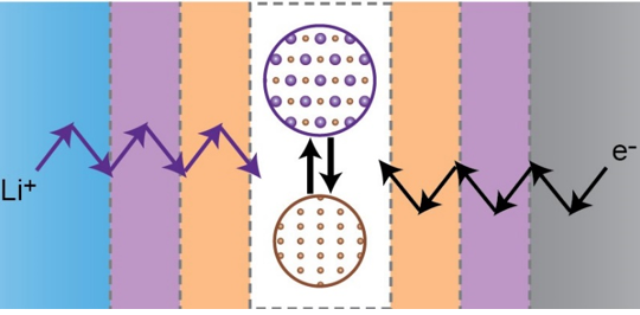
What is the scientific achievement?
CFN staff and collaborators studied the performance of magnetite (Fe3O4) as an inexpensive, nontoxic battery material. Batteries made using magnetite can have high capacities, but unfortunately their capacity fades with battery cycling. The team combined in-situ transmission electron microscopy and synchrotron X-ray absorption spectroscopy to understand the origins of this capacity —directly observing the accumulation of obstacles to electron transport in the magnetite material.
Why does this achievement matter?
In-situ transmission electron microscopy allowed direct observations of electrode structural changes in real time. Understanding how kinetic barriers are linked to capacity fading in materials is important for their future practical implementation.
Tuning Electron Dynamics at 2D Interfaces

What is the scientific achievement?
CFN Users from Columbia University working in collaboration with CFN staff found that intercalating oxygen at the interface between graphene and iridium can change the electronic states of this interface resulting in nearly-flat bands. The scientists used micro-spot angle-resolved photoemission spectroscopy (µARPES) measurements and density-functional-theory calculations to explain the mechanism by which ordered oxygen induces a nearly-flat band structure in graphene.
Why does this achievement matter?
Controlling the electronic states of 2D interfaces will enable new opportunities to engineer band structures and electronic properties in graphene and other 2D materials.
Looking at Atoms to Make Cleaner Fuels from Petroleum
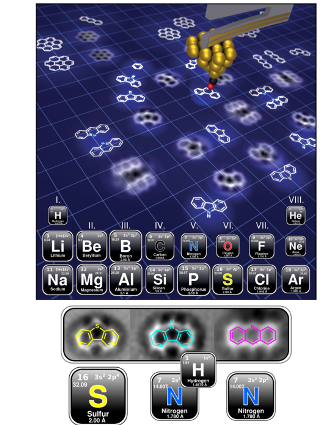
What is the scientific achievement?
CFN staff and users from ExxonMobil have developed a new approach to identifying heteroatoms, like nitrogen and sulfur, commonly found in aromatic hydrocarbon molecules. The team used non-contact atomic force microscopy (nc–AFM) measurements to determine the chemical structure of molecules that can be found in complex mixtures of crude oil.
Why does this achievement matter?
NOx and SOx are two major pollutants that result from the combustion of fossil fuels. Straightforward and robust methods for identifying nitrogen- and sulfur-containing hydrocarbon molecules can improve methods to produce cleaner fuels from crude oil.
Artificial Intelligence for Smarter & Faster Research
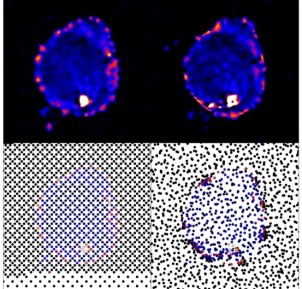
What is the scientific achievement?
A collaborative team of DOE scientists developed a new artificial intelligence method for autonomous experimentation. Their versatile algorithm was integrated into an X-ray scattering instrument and deployed to study nanomaterials without need for human interaction.
Why does this achievement matter?
Machine-guided scientific studies can liberate human scientists from micro-managing experiment details, allowing them to focus instead on understanding the scientific meaning of the results. The methods in this work demonstrate the ability of autonomous methods to achieve high-fidelity searches of experimental problems more efficiently than traditional approaches.
Manganese Nanocatalysts Provide a Fuel Cell Boost
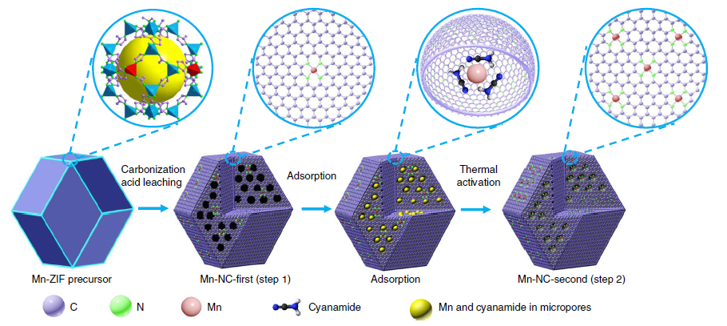
What is the scientific achievement?
CFN and CNMS users from the University of Buffalo led a collaborative study of a new manganese catalyst that significantly enhances the important oxygen reduction reaction in fuel cells. The catalyst enables a large half-wave potential of 0.80 V and remains stable in acidic environments in which fuel cells operate. Mechanistic studies identify the 4-electron pathway responsible for the enhanced performance. The catalyst structure — atomically dispersed MnN4 embedded in graphitic carbon, was established by multimodal X-ray absorption spectroscopy and atomic resolution electron microscopy.
Why does this achievement matter?
Discovery of catalysts that are free from platinum group metals is necessary for wider adoption of fuel cell technologies. The catalyst structure and reaction mechanism identified here provide clues for progress on this important goal.
Freeze Frame: Scientists Capture Atomic-Scale Snapshots of Artificial Proteins

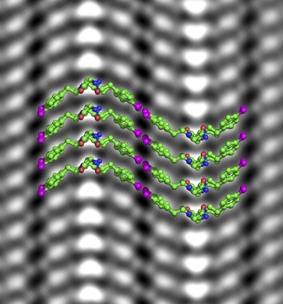
Berkeley Lab scientists adapt microscopy technique to build and image peptoid nanosheets with unprecedented atomic precision
Protein-like molecules called “polypeptoids” (or “peptoids,” for short) have great promise as precision building blocks for creating a variety of designer nanomaterials, like flexible nanosheets – ultrathin, atomic-scale 2D materials. They could advance a number of applications – such as synthetic, disease-specific antibodies and self-repairing membranes or tissue – at a low cost.
To understand how to make these applications a reality, however, scientists need a way to zoom in on a peptoid’s atomic structure. In the field of materials science, researchers typically use electron microscopes to reach atomic resolution, but soft materials like peptoids would disintegrate under the harsh glare of an electron beam.
Now, scientists at Berkeley Lab have adapted a technique that enlists the power of electrons to visualize a soft material’s atomic structure while keeping it intact.
Nanodiamonds Shine with Single Photons
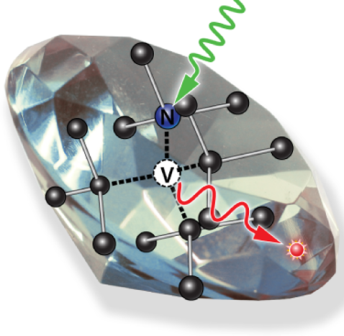
What is the scientific achievement?
Scientists from MIT and CFN researchers have developed a new, scalable approach to creating highly-uniform single-crystal nanodiamonds. The team combined self-assembled nanopatterning with plasma etching for precise synthesis of large quantities of 30 nm diameter nanodiamonds. Photoluminescence measurements demonstrated single-photon emission from single nitrogen vacancy centers located within the nanodiamonds.
Why does this achievement matter?
Solid-state defects are a leading material system candidate for quantum communication and sensing. Single optical defects in nanodiamonds have potential as a sensor platform with unparalleled sensitivity.
Platinum-free Nanocatalysts Provide a Fuel Cell Boost
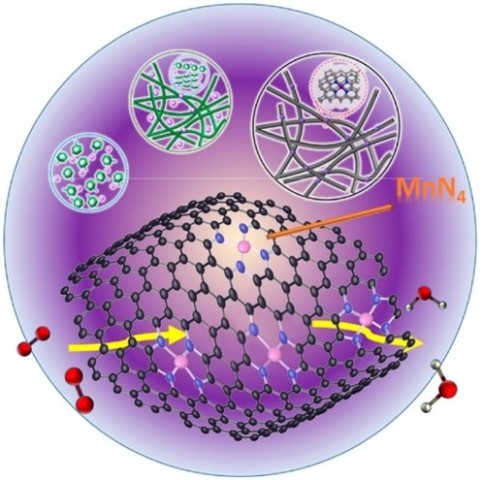
What is the scientific achievement?
A CFN user project investigated Mn- and N- doped catalysts (Mn-N-C) for the oxygen reduction reaction in fuel cells using combined computational and experimental methods. Calculations showed that the Mn-N-C catalyst has the potential to achieve a performance near that of a Pt catalyst (60 mV lower in terms of half-wave potential). Experiments showed the new catalyst has superior stability over 10,000 cycles compared to an Fe catalyst, degrading 75% percent less even after undergoing twice as many potential cycles.
Why does this achievement matter?
Polymer electrolyte membrane fuel cells have the potential to reduce energy use, pollutant emissions, and dependence on fossil fuels. Efficient, stable catalysts that are free from platinum group metals are key for widespread fuel-cell adoption.
Fighting Cancer with DNA Origami
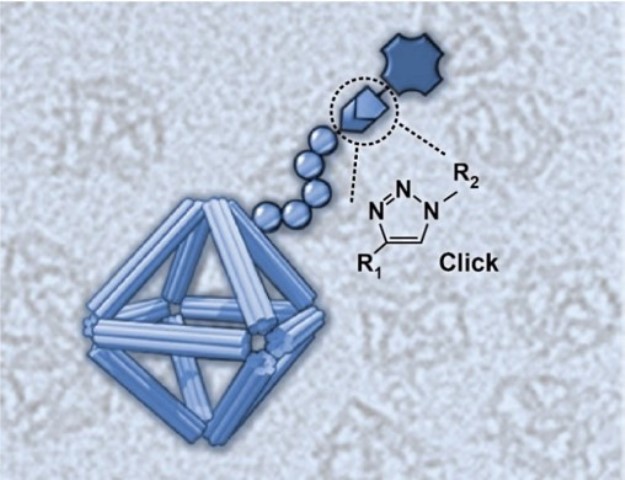
What is the scientific achievement?
CFN scientists and a team of collaborators have devised a new approach for building nanomaterials for use as nanomedicines. The team developed a class of biocompatible molecular coatings and used them to stabilize wireframed DNA origami cages. The coatings give the structure multifunctionality and environmental stability. In this work, the researchers showed that the designed nanomaterials are capable of carrying an anticancer drug and delivering medicines with a controllable release.
Why does this achievement matter?
Although DNA nanotechnology provides a toolkit for creating programmable nanostructures with potential for biomedical applications, a challenge is the limited structural integrity of these materials in complex biological fluids. The molecular coatings developed in this work solve this challenge, paving the way for this approach to be used in drug delivery, bioimaging, and cellular targeting.
Machine Learning Accelerates High-Resolution X-ray Imaging
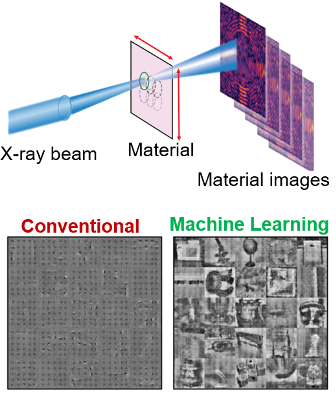
What is the scientific achievement?
A collaborative team from CFN, NSLS-II, and Stony Brook University created a machine-learning algorithm based on a convolutional neural network that accelerates the process of imaging materials with coherent X-rays. This imaging method, called X-ray ‘ptychography,’ is a powerful, high-resolution technique that typically requires long experimental and computational time. The machine-learning algorithm accelerates ptychographic imaging by around 90% based on simulations compared to conventional methods.
Why does this achievement matter?
The speed provided by this new, machine learning-based method makes possible the use of X-ray ptychography for high-resolution studies of beam sensitive materials, and to image in-situ dynamics of nanomaterials in different environments.
Quantum Materials Cloak Thermal Radiation

What is the scientific achievement?
A collaborative team of users, led by University of Wisconsin-Madison, worked closely with CFN staff to show that ultrathin films of samarium nickel oxide can mask the thermal radiation emitted from sources. The cloaking mechanism is due to this quantum material undergoing a unique, gradual insulator-to-metal phase transition across the temperature range of 100 °C and 140 °C — the temperature range of interest.
Why does this achievement matter?
This study shows that quantum materials may be used to manage thermal radiation — important for applications such as infrared camouflage, privacy shielding, and for heat transfer control.
Nanoscale Confinement Speeds Chemical Reactions
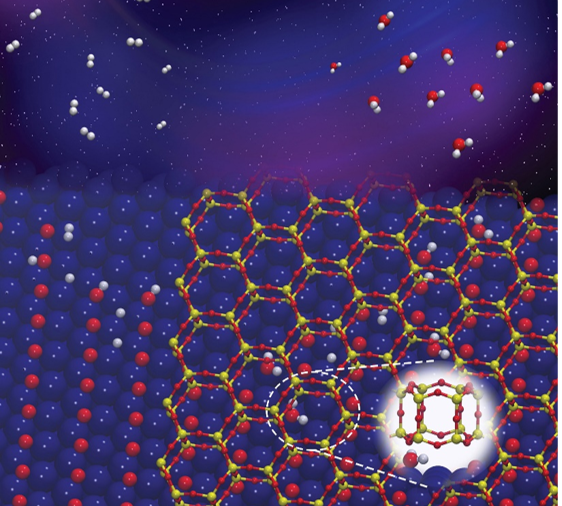
What is the scientific achievement?
CFN scientists discovered how two-dimensional nanoporous silicate covers can accelerate the formation of water from hydrogen gas and oxygen on a metal. The work combines experimental and theoretical methods to demonstrate how the confined nanoscale spaces between the silicate and ruthenium metal accelerate the reaction by lowering the activation energy by 0.38 eV, compared to that of the bare metal surface.
Why does this achievement matter?
The effect of confinement on chemical reactions on surfaces is not well understood. Explaining in detail how confinement influences reactions can lead to new strategies for designing new catalysts and enabling new chemistry.
Better Catalysts to Convert Carbon Monoxide to Fuels
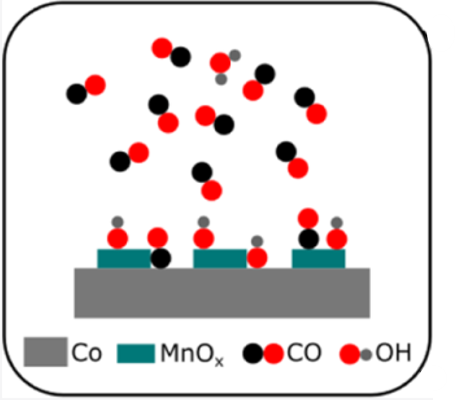
What is the scientific achievement?
Researchers from the Weizmann Institute of Science and CFN showed how manganese oxide enhances the catalytic activity of cobalt, which converts carbon monoxide (CO) and hydrogen to gasoline and diesel fuel. The team used ambient pressure x-ray photoelectron spectroscopy to show that manganese oxide increases CO adsorption on the catalyst surface. Water, a reaction product, competes with CO for adsorption sites.
Why does this achievement matter?
Fischer–Tropsch synthesis is an alternative way to produce petroleum products from nonpetroleum feedstock. This work explains how manganese oxide improves the catalytic activity of cobalt in Fischer–Tropsch synthesis and offers guidance for how to further optimize this process for gasoline production.
Solving the Mystery of Fast Ion Transport in Batteries
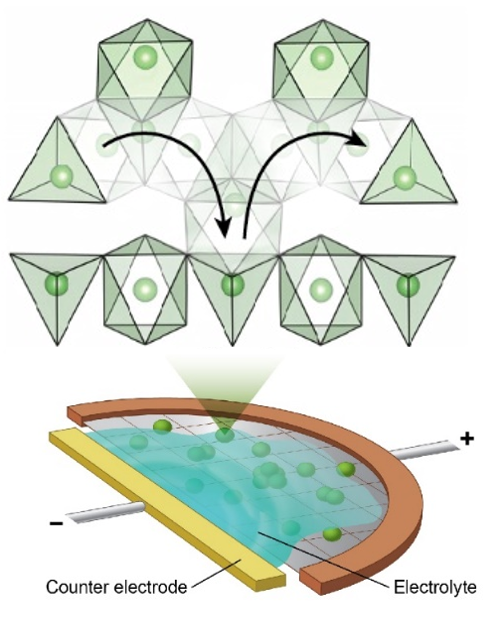
What is the scientific achievement?
A collaborative team of scientists at Brookhaven Lab and UC Berkeley/Berkeley Lab used operando electron microscopy and computational modeling to observe and understand the underlying reasons that lithium titanate battery electrodes can support rapid charging and discharging. Spectroscopy measurements and supporting calculations show that this advantageous anode behavior is enabled by distorted lithium polyhedra in metastable intermediates during battery operation.
Why does this achievement matter?
This work provides a new understanding for designing fast-charging battery materials. A high-rate capability can be designed by accessing metastable structures above the ground state, which can have different kinetic mechanisms.
Machine Learning Deduces Material Structure from X-ray Spectra
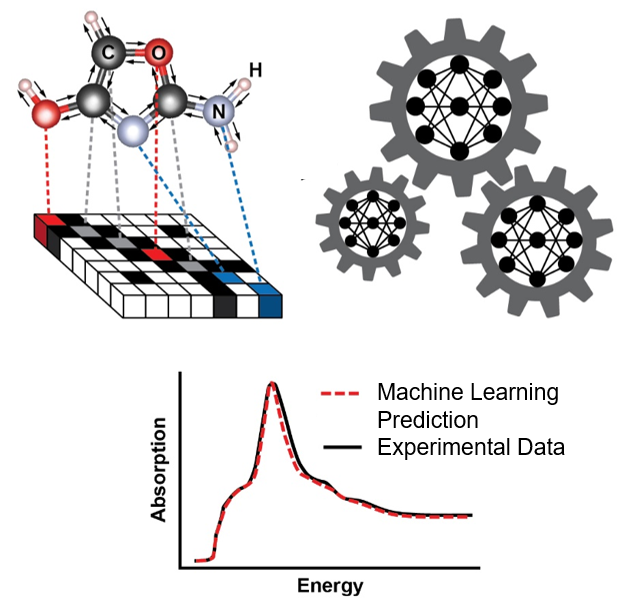
What is the scientific achievement?
A collaborative team from Brookhaven National Laboratory and Columbia University showed that machine learning models can be trained to understand and quantitatively predict relationships between the structure of molecular systems and their x-ray absorption spectra.
Why does this achievement matter?
Unravelling the relationship between the spectral functions of materials and the underlying material structure that leads to these functions is a fundamental challenge in materials science. This study highlights the significant potential for machine learning to extract such key information from x-ray spectroscopy, which can be used for autonomous and machine-guided experimentation.
A New Model for Future Catalyst Design
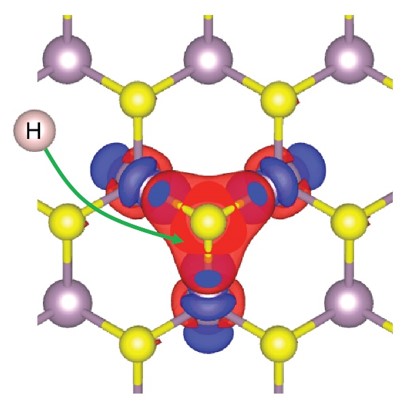
What is the scientific achievement?
CFN scientists have proposed a new theoretical model that explains the increased activity of doped molybdenum disulfide (MoS2) catalysts. The activity of these catalysts depends on the interaction between sulfur atoms on the surface of the material and hydrogen atoms. The model provides a comprehensive understanding of sulfur activation toward hydrogen binding and can be used in future catalyst design. Extensive density functional theory (DFT) calculations were used to carefully validate the model.
Why does this achievement matter?
The work provides a general understanding of the mechanism involved in boosting the activity of MoS2 catalysts doped with metals like nickel, zinc, and cobalt. The activation of inert sulfur atoms could be extended to other materials similar to MoS2 (or transition metal dichalcogenides). More generally, the work could be used to explore other catalytic materials, such as oxides, carbides, and sulfides.
On a Quest to Tailor the Reactivity of Oxide Catalysts
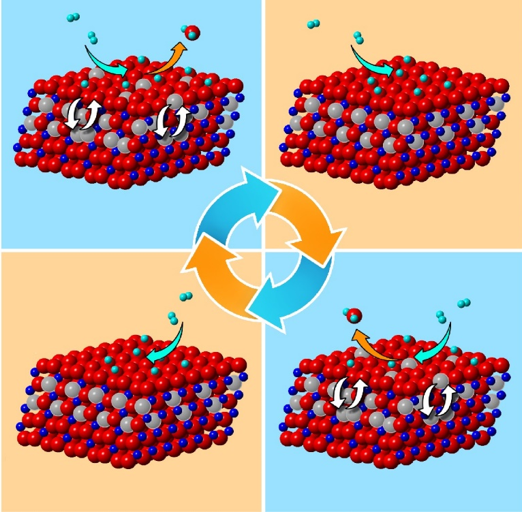
What is the scientific achievement?
CFN users from Binghamton University led a collaborative team studying hydrogen oxidation over copper oxide (CuO) catalyst surfaces by simultaneously resolving atomic-scale structural changes of the surface and subsurface in real time. Cyclic oscillations in the form of ordering and disordering of oxygen vacancies were detected in the subsurface.
Why does this achievement matter?
Differentiating the roles of surface and subsurface states (including nonstoichiometry) in reaction dynamics is important to a wide range of chemical processes involving surface-subsurface mass transport, such as heterogeneous catalysis, oxidation, corrosion, and carburization.
Making 2-D Magnetic Semiconductors Through Substitutions
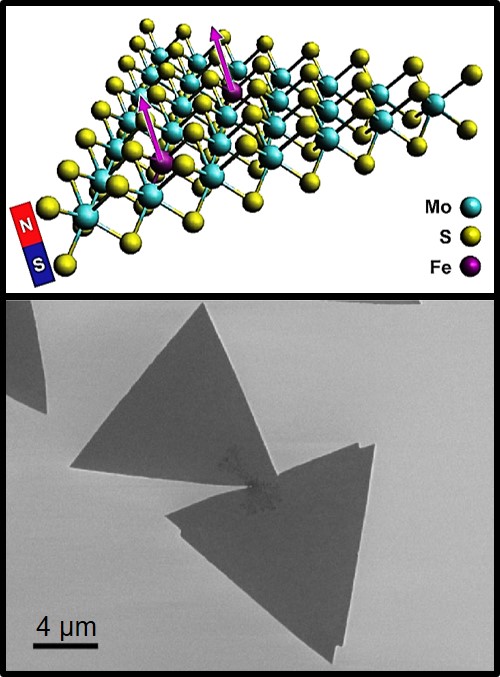
What is the scientific achievement?
A collaborative team of researchers, led by CFN users from Stevens Institute of Technology, demonstrated room-temperature ferromagnetism in the two-dimensional (2-D) semiconductor molybdenum disulfide (MoS2). Small substitutions of iron (Fe) for molybdenum during the material growth process are sufficient to create ferromagnetism, without detrimentally affecting other properties of the material.
Why does this achievement matter?
Previous efforts to form 2-D dilute magnetic semiconductors through extrinsic doping or bulk crystal growth have produced substandard materials. This work provides new dilute magnetic semiconductor 2-D materials, with opportunities to combine them in multilayer stacks for quantum information science or spintronics.
Nanocomposite Liposomes Kill Cancer Cells
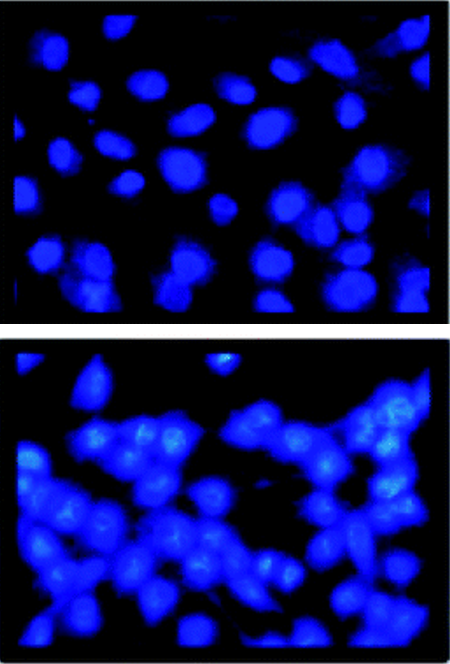
What is the scientific achievement?
CFN users from SUNY Old Westbury developed a nanocomposite cancer medicine by attaching a porphyrin drug to silica and encapsulating it within a nanocomposite liposome. The liposome encapsulant limits drug release to tumor cells, which have an acidic pH, avoiding healthy cells, which are more pH-neutral. The team also showed that singlet oxygen production—used to kill tumor cells—is quintupled in acidic pH compared to neutral pH.
Why does this achievement matter?
Made from phospholipids and biocompatible with cell membranes, liposomes are promising vehicles for nutrient and drug delivery. Lipid selection and modification can be useful for tuning drug targets.
Biofouling Prevention with Nanoscale Polymer Films
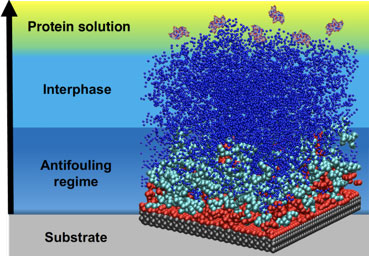
What is the scientific achievement?
CFN users from Stony Brook University and their collaborators from the CNMS at Oak Ridge Lab performed experiments and simulations to reveal a new understanding of the mechanism endowing polymer films with protein-repellent properties. These studies show that polymer films thinner than about 20 nanometers strongly inhibit protein absorption, while thicker films become less effective.
Why does this achievement matter?
Accumulation of biological matter on surfaces, called biofouling, is undesirable in applications ranging from medical implants to aircraft carrier hulls. These results provide new insights into structure-driven protein adsorption under nanoconfinement that will aid in the design of versatile antifouling coatings using common polymers — with important economic and environmental impacts.
Framing up Design of Nanomaterials with DNA

What is the scientific achievement?
CFN scientists developed a platform for self-assembly of ordered, three-dimensional (3D) arrays of DNA frames loaded with nanoscale cargos. Using electron microcopy and synchrotron-based x-ray scattering, the team studied the relationship between the assembled 3D structures and DNA frame designs. They demonstrated use of this platform for design of new optical and catalytic nanomaterials.
Why does this achievement matter?
Organizing materials using cargo-carrying DNA frames decouples the assembly process from the cargo properties (e.g., size and shape) — a long-standing challenge for self-assembly methods. These types of organized nanosystems have appeal for catalytic, optical, energy, and biological applications.
Learning to grow: control of materials self-assembly using evolutionary reinforcement learning

Scientific Achievement
Neural networks were designed to autonomously control experimental parameters, such as temperature and chemical potential, in order to promote the self-assembly of target structures, finding new and nonintuitive pathways to high-quality molecular assemblies.
Significance and Impact
This study is the first to show that self-assembly protocols can be designed autonomously without continuously monitoring the structure during synthesis. This machine learning approach can inform any assembly protocol, including those not well-described by existing theories, such as the crystallization of pharmaceuticals.
Research Details
- The team designed a neural network scheme to control experimental self-assembly parameters using stochastic molecular simulations of prototypical “patchy” particles. Beyond the specification of the target, no human input was required.
- The team compared results from their evolutionary schemes to intuitive cooling simulations, and found that their networks were able to promote the self-assembly of desired structures faster and with higher fidelity.
- In the case where two possible structures have the same energy and are thus equally likely to occur in conventional simulations, the team’s neural networks could reliably promote one polymorph and suppress the other, offering new physical insights into these nonequilibrium processes.
S. Whitelam, I. Tamblyn. “Learning to grow: control of materials self-assembly using evolutionary reinforcement learning.” Phys. Rev. E, 2020. DOI: 10.1103/PhysRevE.101.052604
Lithium as a Photocatalytic Performance Enhancer
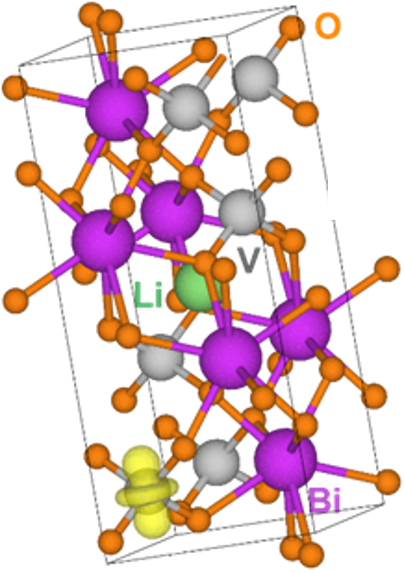
What is the scientific achievement?
Bismuth vanadate (BiVO4) is a promising solar water-splitting material because of its stability in water and ability to broadly absorb sunlight. However, the material is plagued by poor electrical conductivity. CFN staff and collaborators improved the photocatalytic performance of BiVO4 thin-film electrodes by 20% by adding a lithium (Li) dopant via pulsed laser deposition (PLD). The enhanced performance stems from an improved photocurrent output (2.6 mA/cm2), which results from better charge transport. Experimental and theoretical studies verified that the Li dopant acts as a shallow donor to improve the conductivity of BiVO4.
Why does this achievement matter?
This work provides new insight into using Group I elements as low-cost, environmentally friendly dopants to improve electronic transport in metal oxide–based photoelectrodes for solar water splitting.
Catching Light with 3D Hybrid Nanostructures

What is the scientific achievement?
CFN scientists developed a new method to create hybrid optical coatings with tunable antireflective properties; these coatings can also be converted to conductive three-dimensional (3D) nanomeshes. The team combined 3D block copolymer (BCP) self-assembly and heated liquid-phase infiltration of platinum (Pt) into nanostructured organic templates at metal loading fractions 1,500 percent higher (by volume per length) than those for room-temperature infiltration.
Why does this achievement matter?
This liquid-phase infiltration process enables rapid, high loading of metals into unique nanomorphologies. These hybrid nanomaterials have potential as smart optoelectronic coatings, catalysts, and sensors. The infiltration approach uses readily available metal-containing precursors and does not require specialized equipment.
Patching Cracks in Perovskite Solar Cells for Better Performance
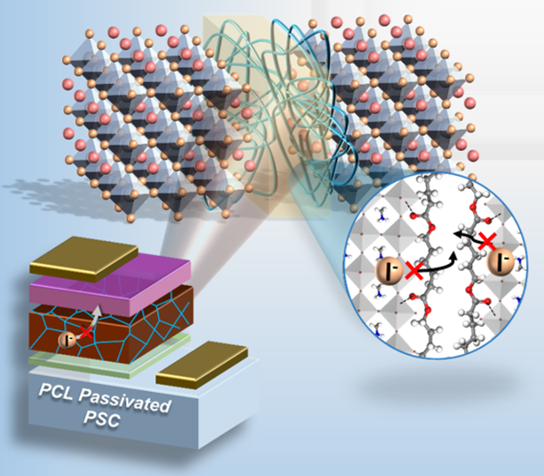
What is the scientific achievement?
A collaborative team of users and CFN researchers demonstrated that the polymer additive polycaprolactone improves the stability of hybrid perovskite solar cells. The polymer passivates grain boundaries in methylammonium lead triiodide perovskite solar cells, suppressing moisture ingression and ion migration. The team demonstrated that solar cells with an initial 20.1% power conversion efficiency retained 90% of their performance after 400 hours of air exposure and 80% after heating for 100 hours at 85°C.
Why does this achievement matter?
This study shows that properly designed polymers can be used to passivate grain boundaries in high-performance, but relatively unstable, perovskite solar cells and provides a potential pathway to practical implementation of these devices.
Taming the photon-magnon interaction

Working with theorists in the University of Chicago’s Pritzker School of Molecular Engineering, Argonne’s researchers have demonstrated a novel approach that allows real-time control of the interactions between microwave photons and magnons, potentially leading to advances in electronic devices and quantum signal processing. Microwave photons are elementary particles forming the electromagnetic waves used for wireless communications. Magnons are the elementary particles forming “spin waves” — wave-like disturbances in an ordered array of microscopic aligned spins that can occur in certain magnetic materials. Microwave photon-magnon interaction has emerged as a promising platform for classical and quantum information processing. This interaction had proved impossible to manipulate in real time. By smart engineering, the team employs an electrical signal to periodically alter the magnon vibrational frequency and thereby induce effective magnon-photon interaction. The result is a first-ever microwave-magnonic device with on-demand tunability. The device can control the strength of the photon-magnon interaction at any point as information is being transferred between photons and magnons. The team’s discovery opens a new direction for magnon-based signal processing and should lead to electronic devices with new capabilities. It may also enable important applications for quantum signal processing, where microwave-magnonic interactions are being explored as a promising candidate for transferring information between different quantum systems.
The Guiding Power of Graphene in Polymer Films
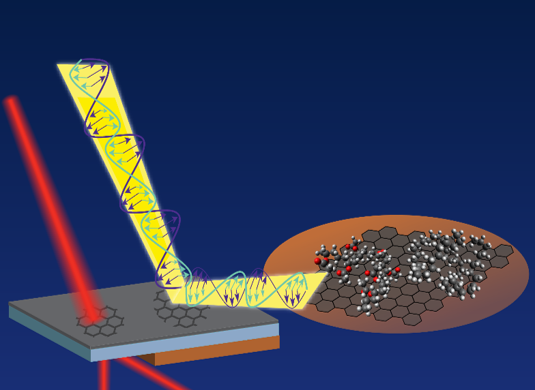
What is the scientific achievement?
A collaborative team of CFN users and staff demonstrated how graphene induces polymer chains to rearrange and create favorable electrostatic interactions. The scientists combined optical transmission, Raman spectroscopy, and infrared reflection absorption spectroscopy to deduce the molecular structure of the polymer-graphene composite films.
Why does this achievement matter?
Conductive transparent film coatings like graphene-polymer composites hold promise for technologies requiring lightweight, strong, and flexible transparent electrodes. This work advances the understanding of graphene-polymer surface interactions and how they can be engineered for use in flexible electronics, energy storage, and biointerfaces.
Using Nanoparticles to Reveal Polymer Nanoarchitectures
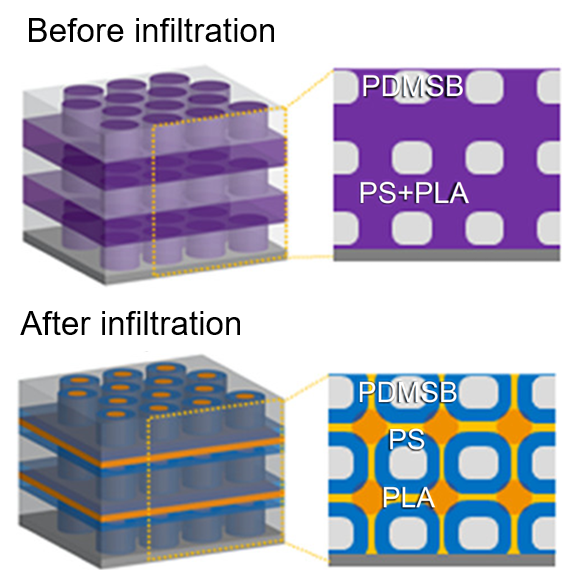
What is the scientific achievement?
Scientists from MIT and CFN revealed the intricate, 3D architectures of self-assembled “three-color” block copolymers by using vapor-phase inorganic infiltration to selectively stain nanostructured polymer thin films with zinc oxide nanoparticles.
Why does this achievement matter?
Complex, 3D ordered architectures of self-assembled block copolymers hold promise for nanofabrication and lithography but are challenging to resolve because of the low contrast between phase-separated blocks. Vapor-phase block-selective infiltration is an effective alternative to slow, toxic, conventional staining for resolving these nanoarchitectures. This work also provides a new nanofabrication strategy for synthesizing multicomponent 3D nanostructures by assembly.
Designing Polarized Light Emitters with DNA
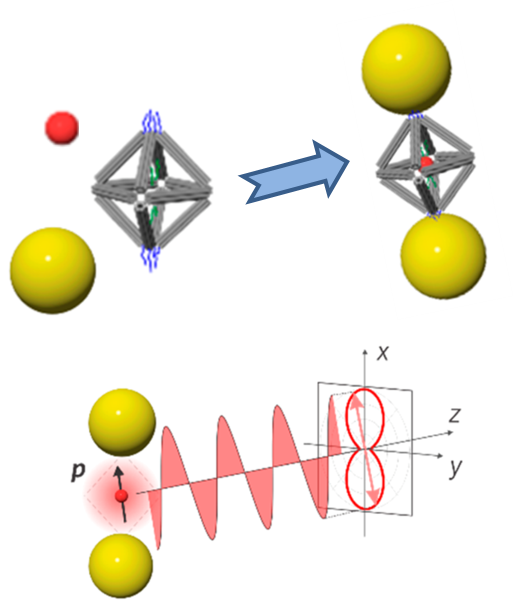
What is the scientific achievement?
CFN researchers and collaborators designed and realized polarized light emitters by self-assembly of nanocomponents. Using DNA origami to mediate optical device assembly, the team programmed coordination of semiconductor quantum dots (QDs) within a DNA octahedron and plasmonic gold nanoparticle (AuNP) antennae at appropriate octahedron vertices. Polarized light emission is due to the metal nanoparticle configuration effect on the quantum dot electromagnetic environment within the cluster architecture.
Why does this achievement matter?
Creating nanoscale polarized light emitters is challenging but needed for communications and information processing. The DNA origami platform is a particularly useful method for programmable design and assembly of such cluster architectures.
Simulations to the Rescue! Designing Better Alkali Catalysts

What is the scientific achievement?
Brookhaven Lab researchers discovered the mechanism that allows a potassium-modified copper nanoparticles to catalyze the conversion of carbon dioxide (CO2) to methanol with enhanced activity, stability, and selectivity. Using density functional theory calculations and kinetic Monte Carlo simulations, they deduced that potassium ions are active centers for selective binding, accelerators for charge transfer, and mediators for electron tunneling.
Why does this achievement matter?
Alkali metals play a critical role in improving catalytic activity and selectivity for many industrial processes. A fundamental understanding of how alkali catalysts enhance reactions allows researchers to design and tune the performance of catalysts beyond the CO2-to-methanol conversion.
Watch-and-Learn How Nanostructured Thin-Films Grow
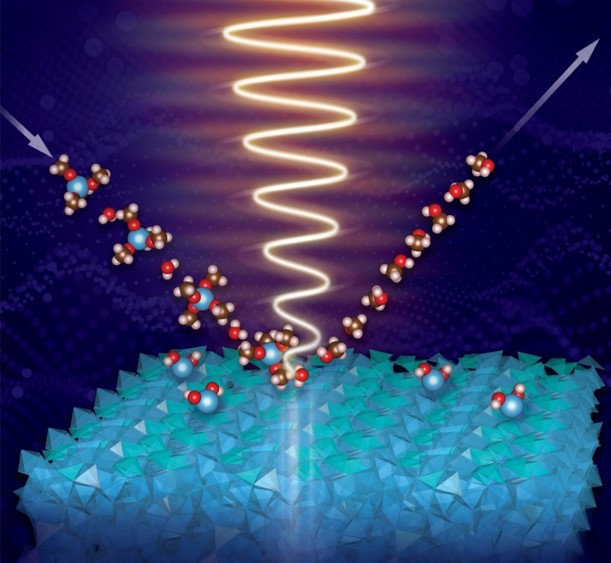
What is the scientific achievement?
By continuously collecting X-ray absorption spectra during atomically-precise film growth, CFN and NSLS-II researchers discovered a new mechanism for how titanium oxide thin films grow on complex, 3D nanoarchitectures. The team developed data analysis tools to deduce a new picture of growth—involving a prenucleation stage, a transition region, and steady state growth—by atomic layer deposition. Loosely-networked titanium polyhedra are present at the surface and interface regions throughout growth.
Why does this achievement matter?
The in situ, X-ray spectroscopy techniques and data analytics methodologies developed in this work are a powerful toolset for understanding & improving vapor-phase film growth, with benefits for microelectronics, catalysis, and solar energy.
Coatings Choose All Sides for a Biomedical Imaging Win
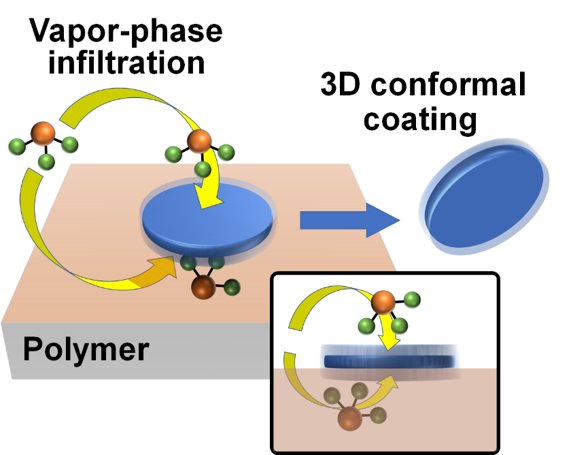
What is the scientific achievement?
Harvard Medical School users and CFN staff developed a new method for coating three-dimensional (3D) laser microdiscs and other nanoparticles with ultrathin metal oxides. Placed on a polymer substrate, the material can be coated on all sides by vapor-phase infiltration synthesis because precursor molecules can diffuse freely through the polymer. The team used this approach to coat laser microdiscs with a 10 nm thick passivating alumina film, allowing the microdiscs to be used in optical cellular imaging in vivo and in vitro.
Why does this achievement matter?
Traditional encapsulation of 3D particles by vapor coating requires large quantities of bulk powders and physical tumbling, making the technique difficult to apply to small amounts of micro- and nanoparticles. The new approach is uniquely applicable to coating even a single nanoparticle.
Having a Ball Designing Nanoarchitectures with DNA
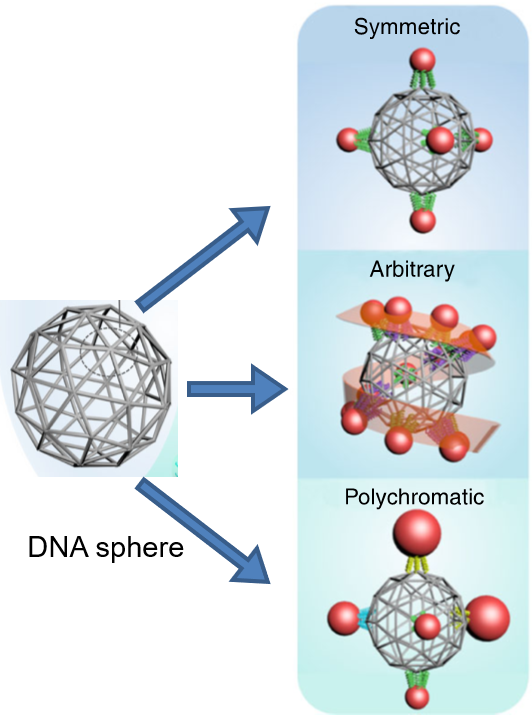
What is the scientific achievement?
CFN researchers created predefined three-dimensional (3D) clusters with complex architectures by programming a sphere-like DNA mesh structure to attach nanoparticles in defined locations. Using this approach, the team programmed the assembly of both symmetric and arbitrary cluster architectures, containing nanoparticles of the same size and different sizes per cluster.
Why does this achievement matter?
Nanoparticle clusters have useful properties for optics, sensing, and information processing, but their precise fabrication by conventional assembly methods is challenging. This work demonstrates that programmable DNA nanostructures provide a universal platform for design and fabrication of cluster architectures with targeted properties.
The Secret to Growing Carbon Nanotube Forests
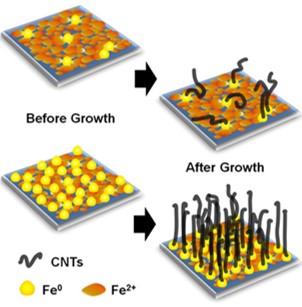
What is the scientific achievement?
A team from the Air Force Research Laboratory and CFN determined the influence of carbon and hydrogen on iron nanocatalysts used to grow “forests” of carbon nanotubes. This work shows that carbon byproducts can efficiently activate the catalysts by reducing them, resulting in the growth of densely packed, tall carbon nanotube forests in high yield.
Why does this achievement matter?
Carbon nanotubes have physical and chemical properties attractive for sensors, energy storage, and composite materials. This study provides a new fundamental understanding of common practices used to synthesize these materials and provides a pathway to improving carbon nanotube densities.
Uncovering Better Catalysts in Confined 2D Spaces
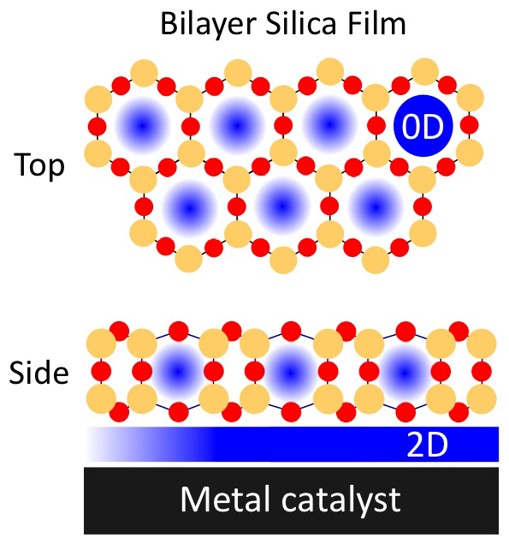
What is the scientific achievement?
CFN scientists demonstrated that nanoporous silica films less than one nm thick boost the activity of a palladium surface for carbon monoxide oxidation, a critical reaction in environmental chemistry. In situ IR & X-ray spectroscopies and mass spectrometry reveal that the film nanostructure disperses CO adsorption and restricts adsorption geometries, enhancing CO conversion and increasing CO2 production by 12 percent, compared to that on palladium alone.
Why does this achievement matter?
Carbon monoxide oxidation converts poisonous CO and O2 to CO2, often with the aid of catalytic palladium. The results reveal the impact of 2D silica films in enhancing metal-catalyzed reactions, which can be applied to other confined reactions and 2D materials.
Learning from Pileups in Li-Ion Batteries
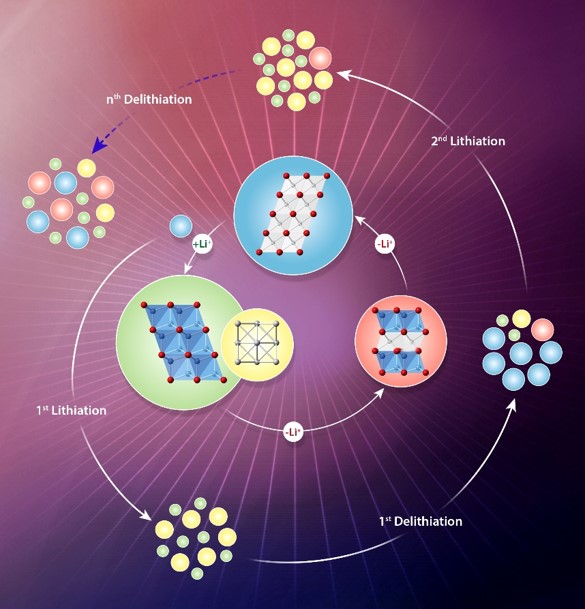
What is the scientific achievement?
A collaborative team of scientists at Brookhaven Lab and Kyungpook National University used transmission electron microscopy and synchrotron x-ray techniques to understand the structural changes in metal-oxide battery electrodes during charging and discharging. They found asymmetric reaction pathways for lithium insertion (charging) and extraction (discharging), with slow lithium-ion diffusion and incomplete lithium extraction during discharge, accelerating the electrode degradation.
Why does this achievement matter?
A detailed understanding of how the structure of conversion-type metal oxide electrodes evolves during battery cycling can be applied to optimize the performance of other conversion-type materials like metal fluorides and sulfides.
A Good Hair Day for Stimuli-Responsive Emulsions
What is the scientific achievement?
CFN users from Binghamton University simulated the interactions between oil-in-water emulsion droplets covered by nanoparticles grafted with hair-like polymers. Their simulations revealed that polymer-grafted nanoparticles are excellent stabilizers of colliding droplets. This stability can be controlled by tuning salt concentration and pH.
Why does this achievement matter?
The ability to stabilize or destabilize emulsions on demand is important for many applications, including enhanced oil recovery, drug delivery, and catalysis. This study provides important insights into designing responsive, switchable emulsions.
A Graded Route to Constructing Stable Batteries

What is the scientific achievement?
A team led by Brookhaven Lab scientists discovered a new way to stabilize high-nickel-content battery cathodes to improve their long-term performance. These materials are attractive for their high storage capacity but suffer cracking upon charge/discharge cycling. Here, the cathode was purposefully designed to have a nickel oxidation state that gradually changed from the surface to the interior. The cathode retained 88 percent of its storage capacity after 100 cycles, compared to 70 percent in a control material.
Why does this achievement matter?
Because of their high energy density, high-nickel-content cathodes have potential to power electric vehicles over much longer distances than supported by current batteries. This study demonstrates the utility of nickel valence gradients for mitigating the poor cyclability of these materials.
Discovering Flat Bands in Unexpected Places
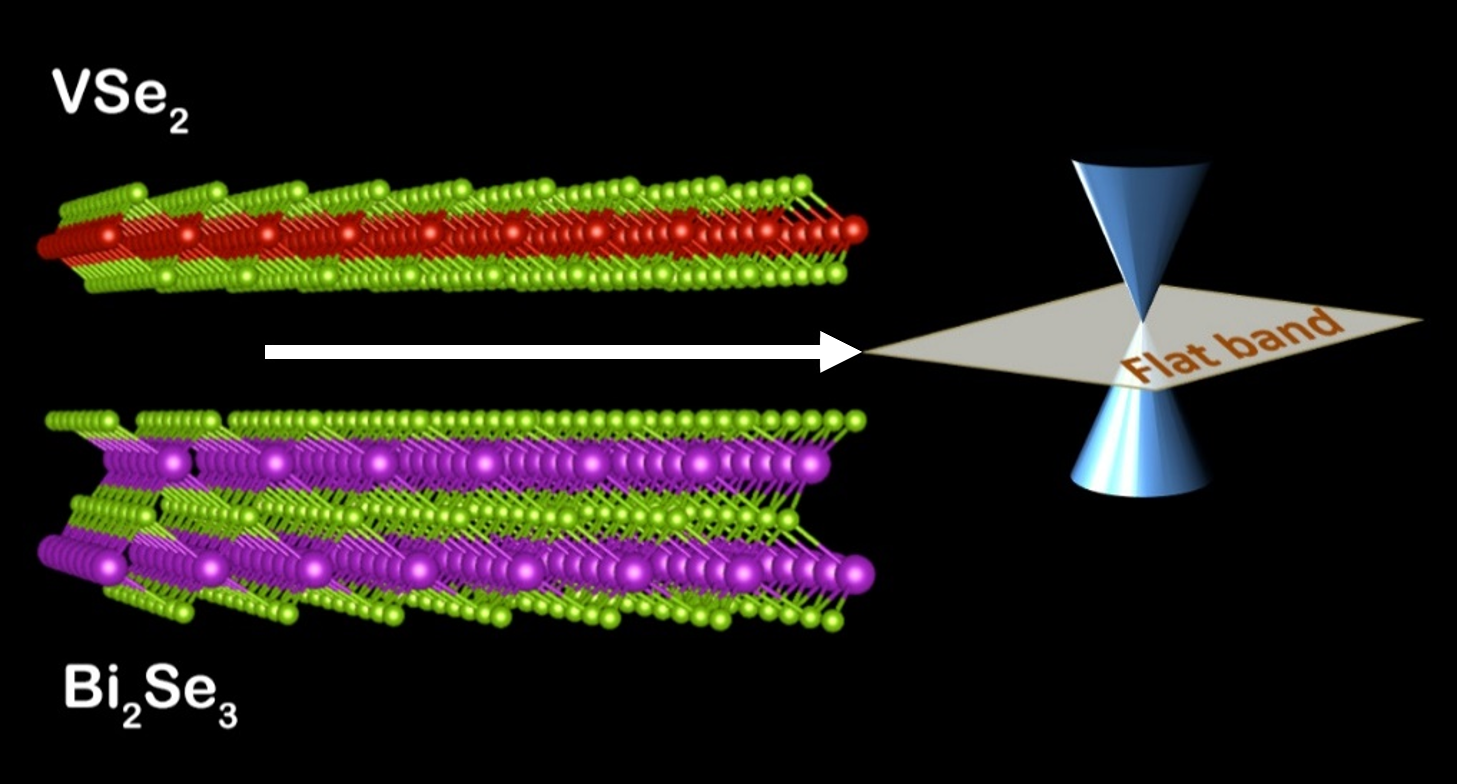
What is the scientific achievement?
A collaborative team of Brookhaven scientists and users made the first experimental observations of the energetically flat electronic band formed at the interface of a heterostructure between vanadium diselenide and bismuth selenide layers. Angle-resolved photoemission spectroscopy (ARPES) measurements confirm the dispersionless character of electronic excitations in the energy band structure.
Why does this achievement matter?
Electronic states in flat energy band structures can possibly support quantum states of matter such as superconductivity, ferromagnetism, and the fractional quantum Hall effect, at higher temperatures, for potential applications in devices for quantum information science. Such flat bands are difficult to realize in material systems.
Disentangling the Roles of Oxygen Vacancies in Solar Water Splitting
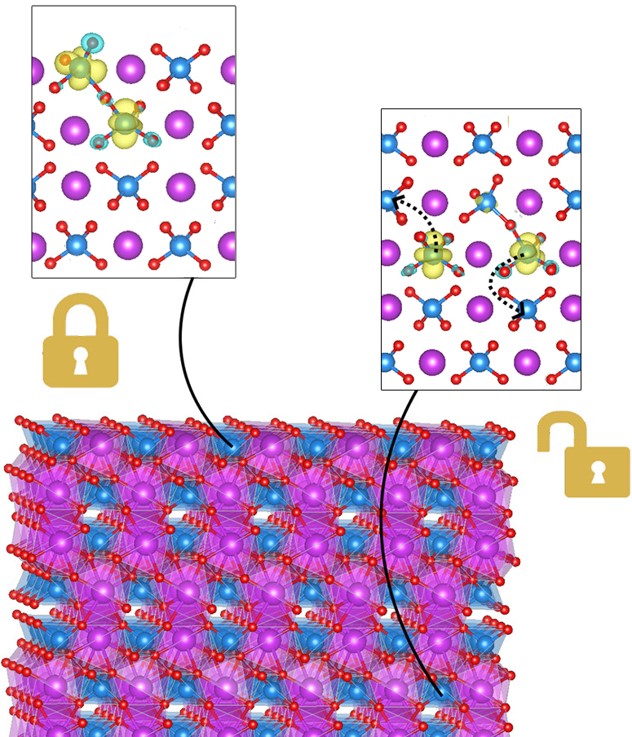
What is the scientific achievement?
CFN users from the University of Chicago led a collaborative team that combined theoretical and experimental studies to understand and improve the solar water splitting performance of a promising oxide catalyst, bismuth vanadate. The team deduced that oxygen vacancies in the bulk contribute to electrical conductivity, but surface vacancies do not. Nevertheless, surface vacancies may be beneficial adsorption sites needed for charge transfer.
Why does this achievement matter?
A holistic understanding of the electronic and catalytic properties of photocatalytic metal oxides such as bismuth vanadate is needed to efficiently generate clean hydrogen fuel from sunlight and water.
Trapping Elusive Radicals in a Metal-Organic Framework
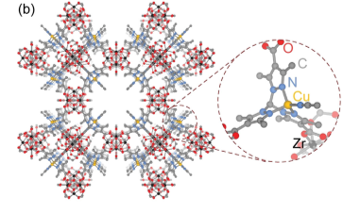
What is the scientific achievement?
CFN users from MIT led a collaborative team who made the first experimental observations of copper-bound anionic hyponitrite (N2O2.− ), an unusual and highly reactive radical. The team trapped the radical using a copper binding site in a metal-organic framework and observed it using several in situ spectroscopy techniques, including x-ray photoelectron spectroscopy at the CFN. They substantiated experimental observations through density functional theory simulations.
Why does this achievement matter?
Anionic hyponitrite is often proposed as a key reaction intermediate in the reductive coupling of nitric oxide to nitrous oxide, but it is extremely challenging to observe. This work demonstrates that metal-organic frameworks can isolate highly reactive species to make them observable. The strategy employed in this work can be used to isolate intermediates in enzymatic catalysis and provide key mechanistic insights.
Peering at Dynamic Perimeters that Promote Catalytic Activity
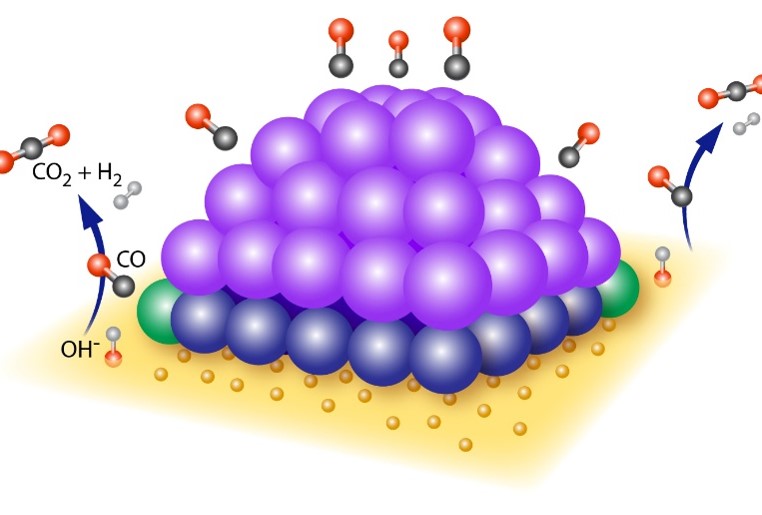
What is the scientific achievement?
CFN users and staff experimentally determined the mechanism by which ceria-supported platinum (Pt) catalysts facilitate the water-gas-shift reaction. Multimodal in situ and ex situ characterization under working conditions illuminate the dynamic changes driving catalytic activity. These measurements showed that the reaction is catalyzed at the perimeter of the Pt-ceria interface at high temperature, with missing oxygen atoms in the ceria also involved in forming the dynamic active site.
Why does this achievement matter?
Oxide-supported precious metal catalysts are of interest for the industrially important water-gas shift reaction to produce hydrogen because of their low-temperature activity, selectivity, and stability. Knowing which sites enable the reaction to proceed can inform the design of catalysts with higher activity at lower cost.
New Model Describes COVID-19 Waves, Plateaus, and Endemic States
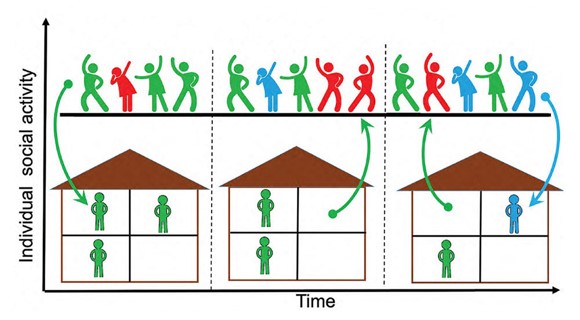
What is the scientific achievement?
CFN staff collaborated with scientists at the University of Illinois Urbana-Champaign (UIUC) to develop a new epidemiological model to explain different and sometimes unexpected outcomes of the COVID-19 pandemic. The team paired a traditional epidemiological model with a stochastic model of variable social activity, which explains both the short-term suppressions of epidemic peaks and the observed, prolonged plateau-like behaviors of COVID-19 cases.
Why does this achievement matter?
The new model is the first to incorporate stochastic social activity into a framework of epidemiological modeling for improved agreement between prediction and reported data, which has been used to support the Government of Illinois and to UIUC with their response to the pandemic.
Adding Up to Create Environmentally Friendly Anti-Corrosion Coatings
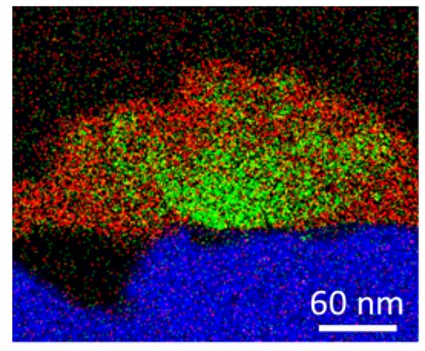
What is the scientific achievement?
CFN users from Stony Brook University, Brookhaven Lab, and Henkel Corporation worked with CFN staff to understand the mechanistic benefits of organic (polyamidoamine) and inorganic (Cu) additives on environmentally friendly, zirconium-based (Zr-based) anti-corrosion coatings. Multimodal characterization by electron- and x-ray techniques characterized the structure and chemistry of the coatings, revealing that optimal amounts of each additive promotes improved morphology, uniformity, and substrate adhesion of the Zr-based coating.
Why does this achievement matter?
Corrosion is destructive to metals and alloys and causes substantial economic, health, and ecological loss. Conventional anti-corrosion coating materials are toxic and carcinogenic. This mechanistic understanding will inform development of environmentally friendly surface-treatment technologies optimized to inhibit corrosion in the automotive, aerospace, and energy storage industries.
Visualizing the Internal Structure and Defects in 3D, Self-assembled Nanomaterials
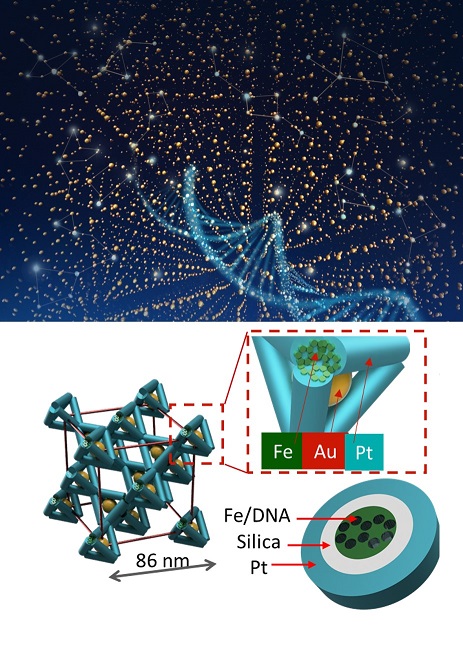
What is the scientific achievement?
A team of scientists from Columbia University, the Center for Functional Nanomaterials, & National Synchrotron Light Source II used advanced X-ray imaging to reveal the structure, composition, and imperfections of 3D nanomaterials designed & synthesized through DNA-mediated assembly and inorganic templating.
Why does this achievement matter?
This work establishes new methods to overcome the difficult challenge of non-destructively verifying the internal structure of complex 3D materials — materials that have promise for catalytic, optical, and energy applications because of their design flexibility.
Pressing Out Clean Interfaces in Layered 2D Devices

Scientific Achievement
CFN collaborators and staff demonstrated systematic fabrication of atomically clean graphene heterostructures with record large dimensions (as large as 7,500 µm2). The team illuminated a new cleaning mechanism, combining mechanical actuation and thermal activation, that achieves atomically-clean 2D layers for use in state-of-the-art devices.
Significance and Impact
This cleaning mechanism is an important tool for developing manufacturing protocols for large and high-quality devices, and obviates additional post-cleaning processes.
Storing Electronic Data in Nanostructured Hybrids
Scientific Achievement
CFN scientists created a hybrid, organic-inorganic polymer and integrated it into a memory device, showing the ability to tune device switching voltage, off-state current, and stochastic variation by controlling the amount of infiltrated inorganic material.
Significance and Impact
This work demonstrates control of device switching parameters and reduced stochastic switching, which are critical technology issues for resistive random-access memory (RRAM). These devices show promise as multi-level memories, which are essential elements in neuromorphic computing.
“No Linker” Single-Molecule Circuits

Scientific Achievement
A team of CFN users and staff formed single molecule circuits with single metallocenes and gold electrodes. They found that barrel shaped metallocenes bind to gold without need for chemical link groups, and exhibit extended conductance plateaus. Metallocenes bind more strongly to a dull tip, compared to a sharp one.
Significance and Impact
Understanding how metallocenes form junctions differently from the more widely studied, rod shaped molecular wires can allow for further manipulation and tunability of these single-molecule circuits.
Unexpected Kinetics in Clean Energy Catalysts
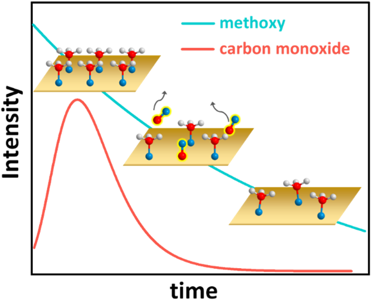
Scientific Achievement
Researchers at the Weizmann Institute of Science and CFN discovered that methanol to hydrogen conversion using copper catalysts displays kinetic behavior that deviates from well-established models, which is explained by measurements of intermediates in the reaction pathway.
Significance and Impact
This work illuminates a better understanding of interactions between methanol vapor and copper surfaces, which is important for the development of tunable copper catalysts for hydrogen production.
More Hydrogen from “Hot” Water Splitting
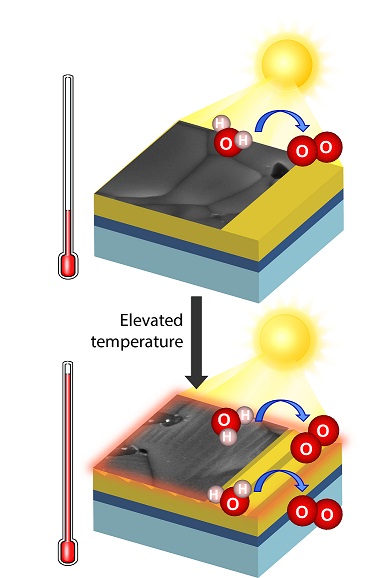
Scientific Achievement
CFN researchers demonstrated that elevated electrolyte temperatures increase the activity of a bismuth vanadate electrode for solar hydrogen generation by photoelectrochemical water splitting by 40 percent, due to more efficient charge carrier separation and a favorable semiconductor surface reconstruction.
Significance and Impact
This work provides insights on the effect of operating temperature on photoelectrode surface morphology and provides a new understanding of scavengers commonly used in photoelectrochemical studies.
A New Quantum Algorithm for Quantum Chemistry
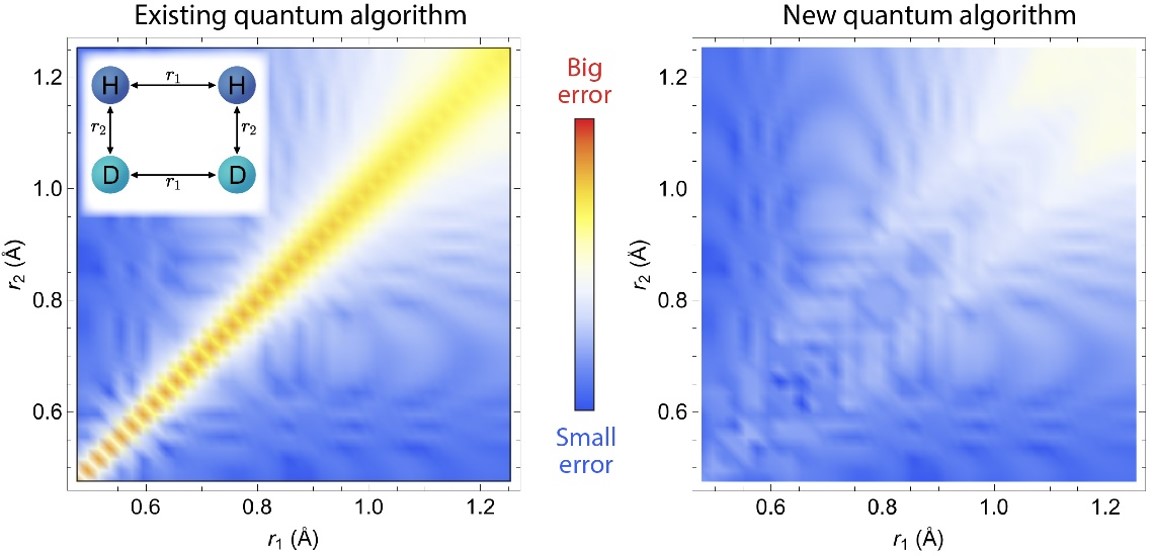
Scientific Achievement
Through the BNL-led Co-design Center for Quantum Advantage (C2QA), a team of scientists at Stony Brook and CFN proposed a new quantum algorithm to accurately solve total energies of molecules.
Significance and Impact
This work will find application in quantum chemistry, such as predicting reaction energies and lowering barriers for future quantum devices by tackling the limitations caused by energy level crossings and energy gap closing.
All-Solid-State Batteries

In Advanced Materials article, "Stable Anode-Free All-Solid-State Lithium Battery through Tuned Metal Wetting on the Copper Current Collector," (Adv. Mater. 8/2023) CINT scientists and users report a stable anode-free all-solid-state battery (AF-ASSB) with a sulfide-based solid-electrolyte (argyrodite Li6PS5Cl). The battery is achieved by tuning the wetting of lithium metal on the “empty” copper current-collector. In the cover image, the sprinting antelope represent the lithium cations rapidly diffusing through the solid-state sulfide electrolyte with the blood-red sunset behind them. The still water underneath represents the smooth lithium-metal anode, while the mountains represent the dendrites, which the antelope avoid.
Controlling Oxide Catalysts with Peroxides
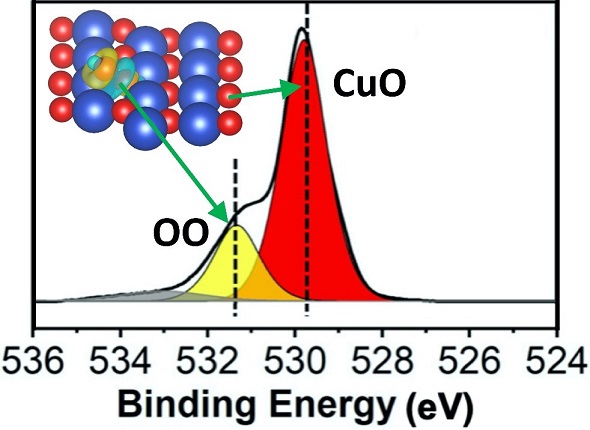
Scientific Achievement
CFN Users at Binghamton University and CFN staff demonstrated they could control the activity and selectivity of an oxide catalyst (cupric oxide, CuO) using adsorbed oxygen.
Significance and Impact
This work provides mechanistic insights into the effect of adsorbed oxygen on the tunability of redox steps for catalytic oxidation reactions, which can play a vital role in clean energy production and environmental remediation applications.
High Throughput and Multimodal Materials Identification
Scientific Achievement
CFN researchers and collaborators employed artificial intelligence (AI) and machine learning (ML) enhanced data analytics and theory, along with high-throughput synthesis and spatially resolved characterization, to identify the phase composition of complex metal-oxide thin films.
Significance and Impact
This robust approach using multimodal studies and data analytics for high-throughput materials discovery may find application in areas such as microelectronics, sensors, and catalysis.
AI Discovers New Nanostructures Using X-rays
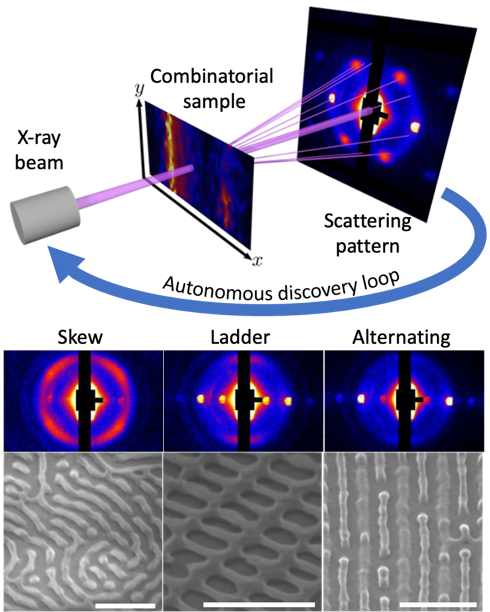
Scientific Achievement
A CFN-led team directed a blend of two self-assembling polymers to form a large library of nanostructures. Microfocused X-ray scattering and machine learning were employed to efficiently explore the library, discovering a nanoscale “ladder” and other previously unknown 3D nanomorphologies.
Significance and Impact
This work shows how combinatorial methods, coupled with autonomous, “self-driving” characterization, accelerates the discovery of self-assembled nanostructures, which have potential applications in microelectronics, membranes, and catalysis.
Explaining the Optical Properties of CdSe Nanoplatelets
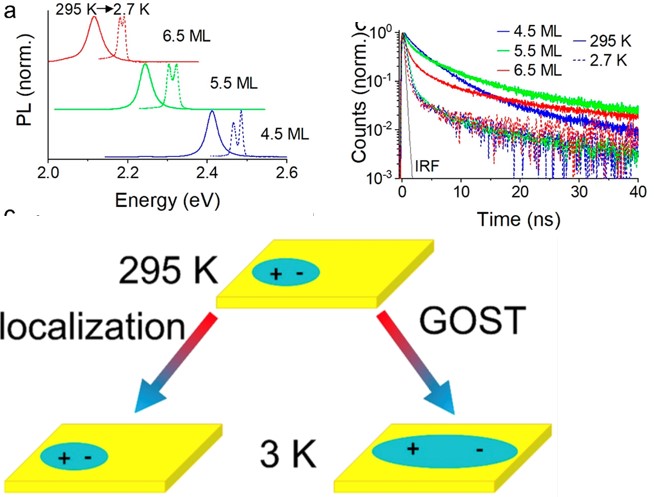
Scientific Achievement
Prior to this work, the excitons in CdSe nanoplatelets (NPLs) were thought to follow the Giant Oscillator Strength (GOST) model, which produces a large increase in exciton size at low temperature. This work shows that is not the case and the exciton remains highly localized at low temperature.
Significance and Impact
CdSe NPLs show far narrower emission features than colloidal quantum dots, and as a result they have attracted great interest in optical applications such as for light-emitting diodes, lasers and quantum optics. This work shows that excitons in NPLs actually remain spatially localized as a function of temperature, while realizing a fast optical emission rate needed for many potential applications.
Research Details
Synthesis of the nanoplatelets was performed at the CNM. All characterization measurements including static absorption, transient absorption, and the optical Stark effect were performed at the CNM between room temperature and 3 K on a series of nanoplatelets.
Characterization of just one atom using synchrotron X-rays

In a Nature paper, scientists report that they have determined both the elemental type and chemical state of single iron and terbium atoms using synchrotron X-rays.
Scientific Achievement
For the first time since the discovery of X-rays in 1895, x-ray spectroscopy has been successfully performed on a single atom. Elemental types and chemical states of an iron and a terbium atom were simultaneously determined using synchrotron X-rays.
Significance and Impact
This work connects synchrotron X-rays to quantum tunneling process thereby enabling simultaneous detection of elemental and chemical states of an atom. Such ability may transform how scientists detect materials and impact research areas that include environmental, medical, and quantum information science.
Research Details
- Research was performed at the XTIP beamline at the Advanced Photon Source and the Center for Nanoscale Materials.
- Fe and Tb atoms were inserted in respective molecular hosts and the X-rays signatures were measured at 30 K using SX-STM.
Understanding the Tantalizing Benefits of Tantalum for Qubits
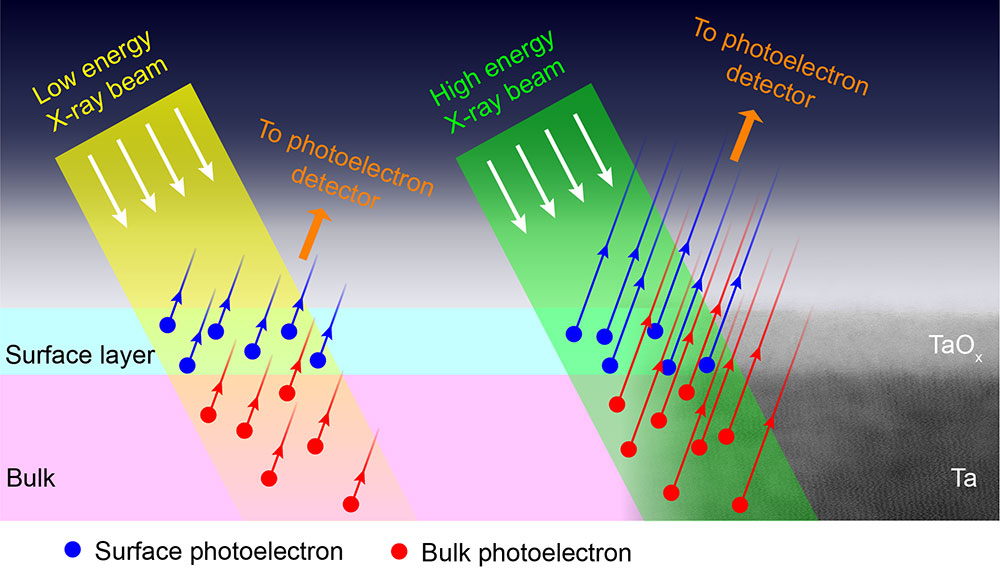
Scientific Achievement
CFN users collaborated with CFN staff on a study to correlate tantalum surface oxidation states with coherence losses in superconducting qubits. Developing a variable energy X-ray photoelectron spectroscopy methodology was key to revealing the depth profile of tantalum oxidation.
Significance and Impact
Tantalum is a promising material for superconducting qubits and has enabled record-long coherence times. Understanding the underlying reasons for qubit decoherence can yield material design pathways to ever better qubit performance.
In Fuel Cell Catalysts, Three Elements are Better than One

Scientific Achievement
CFN staff and users from Binghamton University co-led a study resulting in a new method to synthesize monodisperse, ternary (Pt-Fe-Ni) colloidal catalysts and measure their performance. These trimetallic materials showed superior mass (2.8x) and specific (5.6x) activities toward the oxygen reduction reaction (ORR) compared to the benchmark platinum catalyst, and retain performance after 10,000 voltage cycles in alkaline solution.
Significance and Impact
This synthetic strategy paves the way for improving reactivity for a broad range of electrocatalytic applications, including fuel cells and water electrolysis.
Ultralong coherence of an electron charge qubit
[1].png)
In a Nature Physics paper, scientists report on an electron charge qubit with record long coherence time and strong coupling of two qubits.
Scientific Achievement
The experimental realization of electron charge qubits with a record long coherence time of 0.1 ms and the simultaneous strong coupling of two qubits with a common resonator.
Significance and Impact
This work demonstrates single electron qubits trapped on solid neon as a new promising platform for universal quantum computing.
Research Details
- The quantum coherence of the electron on Ne qubits is significantly improved by Ne annealing, trapping potential stabilization, and the charge-noise-insensitive (sweet) spot qubit operations.
- Ultralong relaxation time and coherence time, 𝑇1,𝑇2𝐸~0.1 ms with high-fidelity single-qubit gates and single-shot readout, outperforming all existing charge qubits to date.
- The dilution refrigerator system at the Center for Nanoscale Materials at Argonne National Laboratory was used.
Coating for Ni-rich oxide cathode yields fast charging and long-term cycling
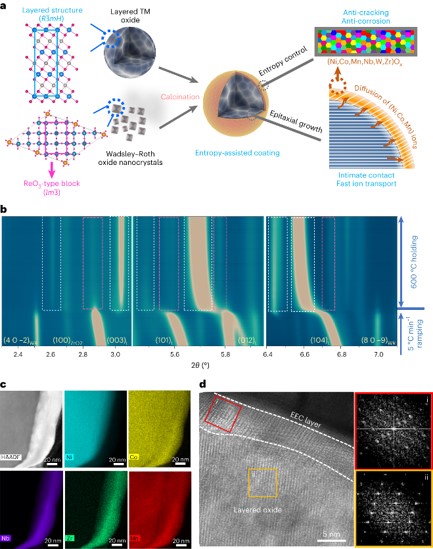
Scientific Achievement
An epitaxial entropy-assisted coating (EEC) was developed for LiNi0.9Co0.05Mn0.05O2 (Ni90) cathodes. The resulting high anti-cracking, anti-corrosion tolerances, and fast ionic transport improved the charging/discharging capability, temperature tolerance, and thermal stability of these Ni-rich cathodes.
Significance and Impact
This coating strategy paves the way for developing fatigue-free Ni-rich cathodes to develop fast-charging and long-lasting lithium-ion batteries with high power and energy.
Research Details
Decision trees plus AI equal discovery of boron metastable phases
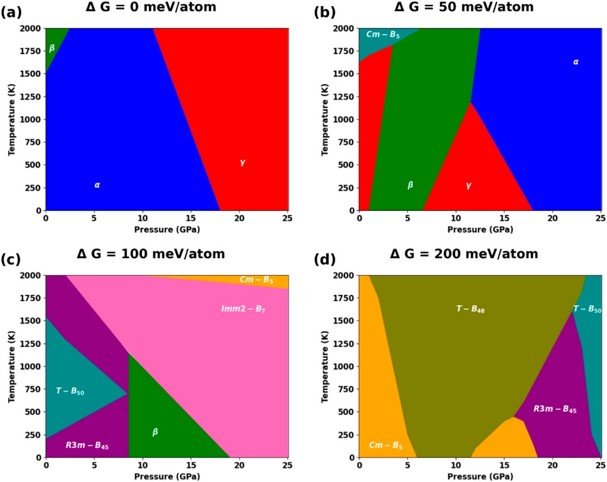
Scientific Achievement
A workflow that uses reinforcement learning coupled with decision trees was developed to search for stable and metastable boron phases.
Significance and Impact
This new framework for representing metastable boron phases offers valuable insights for experimental design, streamlining the discovery process of these phases, renowned for their attractive properties absent in stable phases.
Research Details
Patterning the Future of Biomineralization with Block Copolymers
Scientific Achievement
Users from the University of Washington collaborated with CFN staff to use block copolymer patterns to facilitate template formation of peptide-based nanostructures, which influence the growth of enamel-like calcium phosphate.
Significance and Impact
Supramolecular templates play a crucial role in directing the growth of biominerals such as tooth enamel. This work provides a potent biomimetic platform to understand such templates.
Nanosheets Increase Perovskite Solar Cell Efficiency and Stability
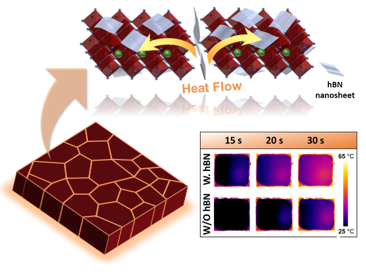
Scientific Achievement
CFN scientists and users from Stony Brook University collaborated to discover that adding 2D hexagonal boron nitride nanosheets to perovskite solar cells expedites crystallization and grain growth, improving the cells' efficiency and stability in air.
Significance and Impact
Improving solar cell efficiency from 17.4% to 19.8%, with 90% efficiency post-500 hours in ambient air, shows the potential of hBN additives in organic and perovskite device design.
Solar-Powered Chlorine Production: A Sea Change in Water Splitting
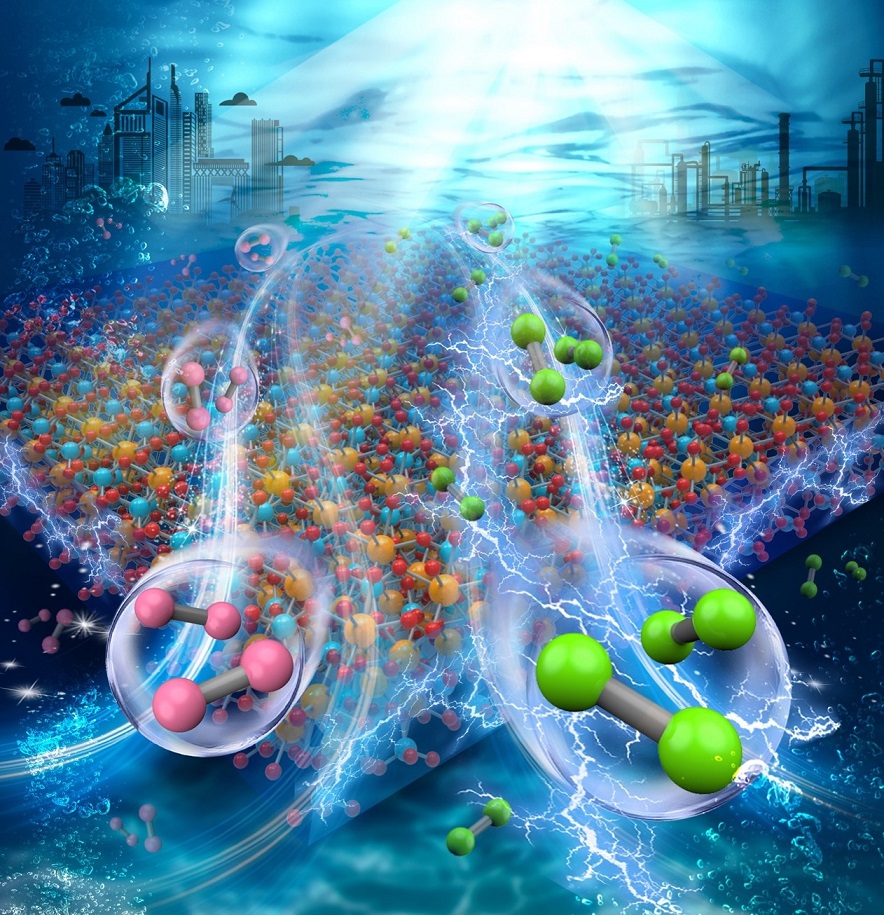
Scientific Achievement
CFN researchers demonstrated a method to generate chlorine and hydrogen from seawater by solar water splitting, using bismuth vanadate and a cobalt oxide co-catalyst. The approach bypasses the bottleneck of the inefficient oxygen evolution reaction.
Significance and Impact
This approach has potential to reduce the energy footprint of the chloralkali industry and repurpose oceanic chlorine for clean energy production, while also providing clean hydrogen fuel.
Novel technique reveals the origin of structural degradation in battery materials
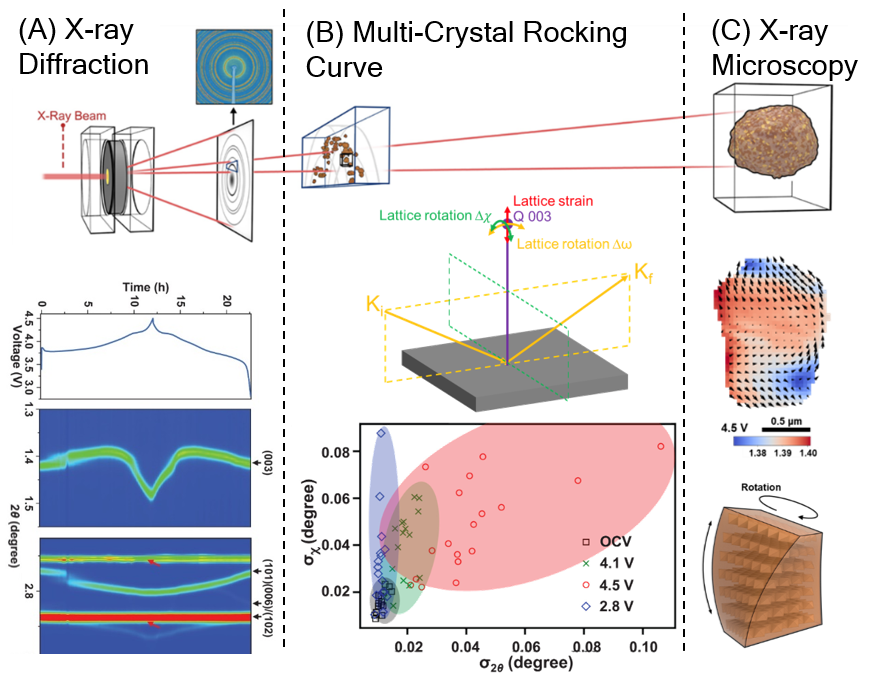
Scientific Achievement
A new technique capturing both the statistical and individual lattice distortion has bridged the information gap between x-ray diffraction and x-ray microscopy.
Unrecoverable lattice rotation was identified as the root cause for structural degradation induced capacity decay in single crystal nickel-manganese-cobalt (NMC) cathode materials.
Significance and Impact
The new technique revealed the origin of structural degradation in Li-ion battery cathode materials, the understanding of which can lead to better capacity retainment and longer battery life.
Research Details
- Development of a new x-ray diffraction technique to study the lattice distortions in an ensemble of particles.
- Discovery of lattice rotation as a primary factor initiating structural degradation in single-crystal cathode particles.
Atomic-Resolution Imaging of Bulk Samples by Scanning Electron Microscopy

Scientific Achievement
Center for Functional Nanomaterials and Brookhaven National Laboratory researchers demonstrated atomic-resolution secondary electron imaging in bulk crystalline samples as thick as 18 µm, using an aberration-corrected scanning transmission electron microscope.
Significance and Impact
This breakthrough enables high-resolution atomic-scale imaging of bulk materials without the need for ultra-thin specimens, which are typically required for electron microscopy at this resolution.
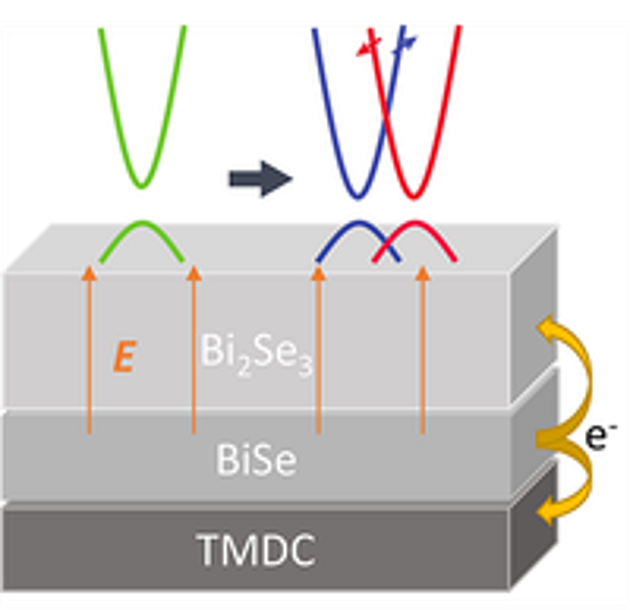
Topological Breakthroughs: Tuning Quantum States in 2D Heterostructures
Scientific Achievement
Through first principles calculations, CFN staff & collaborators engineered topological properties in 2D Bi2Se3/BiSe/transition metal dichalcogenide heterostructures through interface charge transfer, which creates Dirac surface electronic states.
Significance and Impact
This work presents a new method to control topological properties in 2D materials, with promise for designing better optimized electronic materials for quantum devices.
Transforming Polymer Membranes for Better Hydrogen and CO2 Separation
Scientific Achievement
CFN staff and collaborators showed that sequential exposures of polybenzimidazole membranes to trimethylaluminum and water vapor enhance H2 permeability (by 270%) and H2/CO2 selectivity (by 30%), improving utility for separations and blue H2 production.
Significance and Impact
This work demonstrates a scalable strategy to nanoengineer polymeric membranes for improved gas separation performance, crucial for clean H2 production and carbon capture.
Data-driven approach to extract dynamics from coherent X-ray data
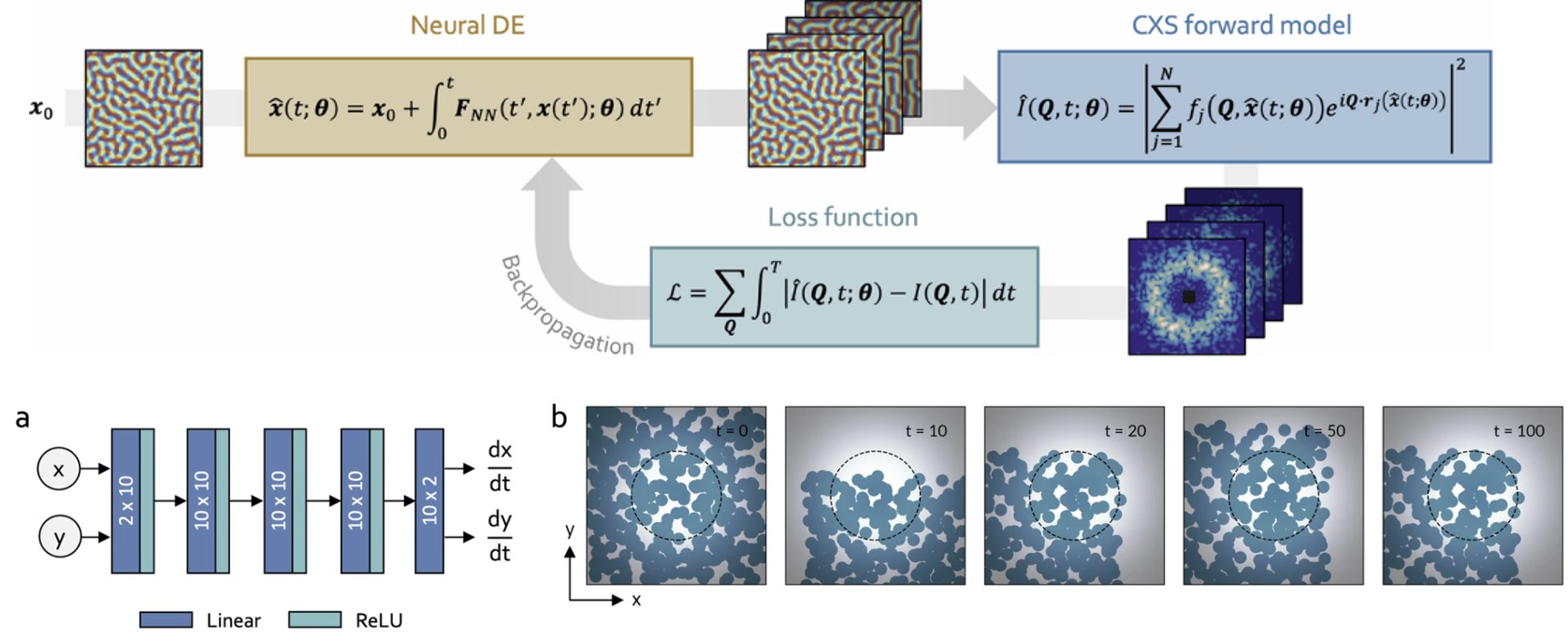
Scientific Achievement
A data-driven platform based on scientific machine learning has been developed to extract complex, real-space dynamics from time-resolved coherent X-ray scattering (CXS) measurements. These data enable estimation of long-term dynamics well beyond the maximum observation time.
Significance and Impact
The platform allows visualizing previously inaccessible dynamics based on a few initial high-fidelity measurements followed by fast scans to infer dynamics.
Research Details
Lighting Up PFAS: Rapid Detection with Fluorescent Nanosensors
Scientific Achievement
CFN users collaborated with staff to demonstrate fluorescent nanosensors using hexanuclear Cerium-oxo clusters (Ce-Gly) for rapid, one-step detection of long-chain carboxylated per- and polyfluoroalkyl substances (PFAS) in environmental samples.
Significance and Impact
This method provides a cost-effective and efficient alternative for PFAS detection, which is critical due to the harmful effects of PFAS on health and the environment. The approach offers potential for widespread environmental monitoring.
Solvent Vapor Takes Nanopore Fabrication to New Heights
Scientific Achievement
CFN and Columbia researchers developed a robust protocol for fabricating highly ordered arrays of vertical nanopores in thin films using the self-assembly of a block copolymer (BCP) and solvent vapor annealing (SVA).
Significance and Impact
This method enables precise control over nanopore dimensions and orientation, with potential applications in nanolithography, membrane filtration, and energy storage.
Magnesium Protects Tantalum, a Promising Material for Making Qubits
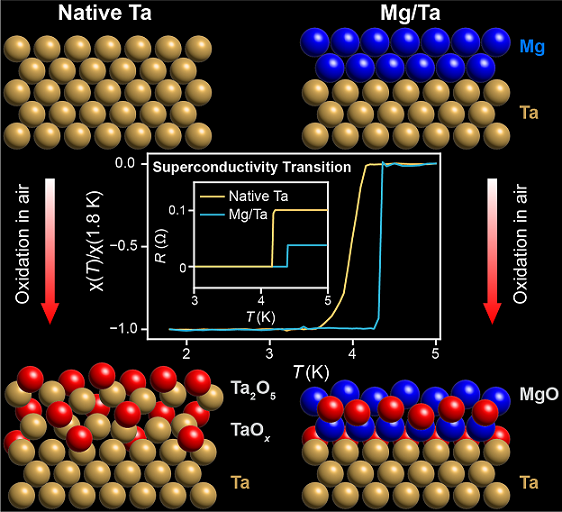
Effective and durable catalysts using abundant materials for hydrogen production

In a study published in ACS Applied Nano Materials, researchers report a novel method to synthesize hybrid 2D FeS/FeS2 nanostructures, a promising low-cost catalyst for next-generation hydrogen production.
Scientific Achievement
A hot-filament metal–chemical vapor deposition method is presented for synthesizing pristine ferromagnetic FeS2 nanoparticles (NPs) and 2D FeS/FeS2 hybrid nanostructures on a carbon substrate. The latter exhibited exceptionally low overpotential and enhanced magnetism.
Significance and Impact
The FeS/FeS2 hybrid catalyst is a promising low-cost alternative to Pt-based catalysts for hydrogen production in next-generation electrocatalytic water-splitting technologies.
Research Details
- Test results showed that the 2D FeS/FeS2 nanostructures exhibited a low overpotential of 40 mV at 10 mA·cm–2 current density and magnetic properties superior to pristine FeS2 NPs.
Enabling preparation of previously inaccessible semiconductor nanocrystals
In a Science paper, researchers report synthesizing III-V semiconductor nanocrystals with possible applications in displays, lasers, photodetectors, and more.
Scientific Achievement
The study demonstrates a new method for synthesizing III-V semiconductor nanocrystals, specifically colloidal quantum dots, using molten inorganic salt solvents. Nanocrystals from binary species, such as gallium arsenide (GaAs), up to pentanary compositions can now be grown.
Significance and Impact
This capability has applications in displays, lasers, photodetectors, solar cells, photocatalysts, photonics, and emerging fields like quantum information science.
Research Details
- The CNM provided femtosecond transient absorption spectroscopy for nanocrystal characterization.
Ruthenium-based single-crystal optical actuator achieves 100% conversion

In a study published in Nature Communications, researchers created a ruthenium-based single-crystal optical actuator achieving 100% reversible conversion between three distinct photoisomeric states enabling precise control over molecular states.
Scientific Achievement
Researchers have developed a ruthenium-based single-crystal optical actuator that can achieve 100% conversion between three distinct photoisomeric states using light and temperature control that is 100% reversible.
Significance and Impact
This work expands the potential of photonic materials, enabling precise control over molecular states for applications in optical data storage, optomechanics and advanced microrobotics.
Research Details
- Variable-temperature light-assisted atomic force microscopy (AFM) and single crystal optical absorption experiments. were performed at CNM.
DOI: 10.1038/s41467-025-56795-w
Directional Phonon Speed in Black Phosphorus for Better Heat Control
In a study published in ACS nano, researchers directly observed that in black phosphorus, phonon speed varies with direction due to structural influences, offering insights into heat control in 2D materials, crucial for efficient electronic devices.
Scientific Achievement
Observation with an ultrafast electron microscope showed that in black phosphorus the speed of phonons varies with direction due to structural influences, highlighting the anisotropic nature and the complex interactions between its lattice structure and thermal transport properties.
Significance and Impact
These results provide new insights into controlling heat in 2D materials, which is crucial for developing more efficient electronic and thermoelectric devices.
Research Details
- The Ultrafast Electron Microscope at the CNM was used to observe how phonons travel through black phosphorus.
- Theoretical calculations were done using Carbon and VASP at the CNM.
Accelerating peptide discovery with machine learning
In a study published in Science Advances, researchers used machine learning to predict unconventional self-assembling peptide materials, refining models over 3 iterations with >250 peptides, accelerating innovation in medicine and electronics.
Scientific Achievement
Researchers successfully used machine learning to accurately predict and discover unconventional self-assembling peptide materials, surpassing traditional design methods.
Significance and Impact
The ability to leverage machine learning to accelerate the discovery of innovative peptide materials has the potential to transform applications like medicine and electronics.
Research Details
- At the CNM, curated a database of > 250 in-house synthesized and spectroscopically characterized peptides.
- Machine Learning refinement occurred over 3 iterations leading to enhanced precision.
Halide segregation boosts all-solid-state lithium-chalcogen batteries
In a study published in Science, researchers discovered a halide segregation mechanism in lithium-chalcogen batteries, enhancing ion transport and stability, improving efficiency and cost-effectiveness.
Scientific Achievement
Researchers discovered a universal halide segregation mechanism at the interfaces of all-solid-state lithium-chalcogen batteries that enhances ion transport and stabilizes the cathode structure, significantly boosting battery performance.
Significance and Impact
This mechanism can enhance the design and manufacturing of all-solid-state lithium-chalcogen batteries, making them more efficient and cost-effective by improving charge transport and mechanical stability.
Research Details
- At the CNM, halide segregated interfacial layer between cathode and solid electrolyte was characterized by low dose cryo-TEM.
- Elemental distribution analysis done at CNM with EDS, EELS.
- Thermal stability study with in-situ annealing.